| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 10, Number 11, November 2019, pages 328-332
The Simultaneity of Coronary Artery Ectasia and Intracranial Arterial Dolichoectasia: A Rare Case Report
Harith Adnan Alatabya, d, George Frega, Rana Alzakharia, Gourg Atteyab, Muhamed Dougarb, Bhavesh Galac, Jay Nfonoyima
aDepartment of Medicine, Richmond University Medical Center, Staten Island, NY 10310, USA
bDepartment of Cardiology, Suny Downstate medical center, Brooklyn, NY11210, USA
cDepartment of Cardiology, Richmond university medical center, Staten Island, NY 10310, USA
dCorresponding Author: Harith Adnan Alataby, Department of Medicine, Richmond University Medical Center, Staten Island, NY 10310, USA
Manuscript submitted November 4, 2019, accepted November 20, 2019
Short title: A Rare Case of CAE and IADE
doi: https://doi.org/10.14740/jmc3390
| Abstract | ▴Top |
In the present study, we report a case of multiple coronary artery ectasias (CAE) and multiple intracranial arterial dolichoectasias (IADEs). A 60-year-old female presented to the emergency department twice with chest pain and mild elevation of troponin and T-wave changes. Peripheral coronary angiography showed severe ectasia and stenosis of certain segments of the left main coronary artery (LMCA), left anterior descending (LAD), first obtuse marginal (OM1), distal left circumflex (LCX), and bilateral subclavian arteries. The patient was treated medically. Two weeks later, she presented with dizziness. Head computerized tomography (CT) angiography showed severe IADE involving the vertebrobasilar system, intracranial internal carotid arteries, and bilateral middle cerebral arteries. No neurovascular intervention was performed due to the complexity of the findings. CAE is an abnormal dilatation of a coronary artery segment of at least 1.5 times the size of a normal coronary artery. The slow flow phenomenon may lead to ischemia and thrombosis, which can result in acute coronary syndrome. IADE comprises a dilatation and elongation of the arteries that affects both the anterior and posterior cerebral circulation, often causing neurological complications such as ischemic stroke, intracranial hemorrhage, or compression of surrounding neural structures. We report this case due to the rarity of coexisting IADE and CAE. A rarefaction of elastic tissue of the media with degeneration of the internal elastic lamina, in addition to matrix metalloproteinase dysfunction, is a common pathological mechanism for this condition. The management of CAE and IADE is mostly conservative, essentially treating the risk factors and administering antiplatelet and anticoagulant agents. In some patients, angioplasty vs. surgical treatment may be applied.
Keywords: Coronary artery ectasia; Intracranial arterial dolichoectasia; Coexistence; Concurrence; Association
| Introduction | ▴Top |
There are a few cases in the literature reporting the association between arterial ectasia in the two most vital organs, the brain, and heart [1, 2]. In the present study, we report a case of multiple hospital admissions on account of transient ischemic attack (TIA) and angina-like symptoms. Following an extensive workup, the patient was found to have multiple coronary artery ectasias (CAE) and aneurysms together with multiple intracranial arterial dolichoectasias (IADEs) and aneurysms. The complexity of this case and the challenge in management rendered reporting of this case essential.
| Case Report | ▴Top |
A 60-year-old female with a medical history of hypertension and hyperlipidemia presented to the emergency department on account of left anterior chest pain that radiated to the left arm. The pain was non-exertional, had been present for a while (not specific), and was mild and intermittent but suddenly grew worse that afternoon. At presentation, she claimed that the pain was no longer present. A review of the systems was negative for any related symptoms. The physical exam was unremarkable, and the vital signs were within the normal range. The electrocardiogram (ECG) showed normal sinus rhythm (NSR), with a heart rate of 70 beats per minute (bpm) and T-wave inversion in V4 and V5. The troponin level was mildly elevated (0.082, 0.084, 0.089, 0.083 ng/mL). Echocardiology showed no left ventricular (LV) wall motion abnormalities and normal overall LV systolic function; the LV ejection fraction was 60%, and the LV diastolic function was indeterminate. The nuclear stress test showed abnormal dipyridamole stress myocardial perfusion imaging, representing moderate inferior and inferolateral ischemia with partial scarring, borderline LV size, and mild LV dysfunction. The patient was a poor candidate for invasive cardiac workup in terms of an angiogram due to severe anemia and was treated medically with aspirin, isosorbide mononitrate, nifedipine, and atorvastatin. The patient’s symptoms disappeared during her stay in the hospital, and she was discharged with a plan to repeat the ECG in 6 - 8 weeks and follow-up with a cardiologist as an outpatient. One month later, the patient was brought to the hospital for evaluation of further chest pain with the same physical and ECG characteristics as the previous episode and minimal elevation of troponin (0.075 - 0.052). Echocardiology showed no differences from the previous. The general condition of the patient was more stable, and the anemia had improved; therefore coronary angiography was performed, the findings of which were: right-side dominance; severe ectasia of the left main coronary artery (LMCA); extreme ectasia of the left anterior descending artery (LAD), with multiple areas of moderate-to-severe distal and apical stenosis; multiple areas of stenosis including the first obtuse marginal artery (OM1), and distal left circumflex artery (LCX); ectopic origin from the left cusp of the right coronary artery (RCA); and total proximal occlusion with the distal vessel receiving collaterals from the left system (Figs. 1, 2).
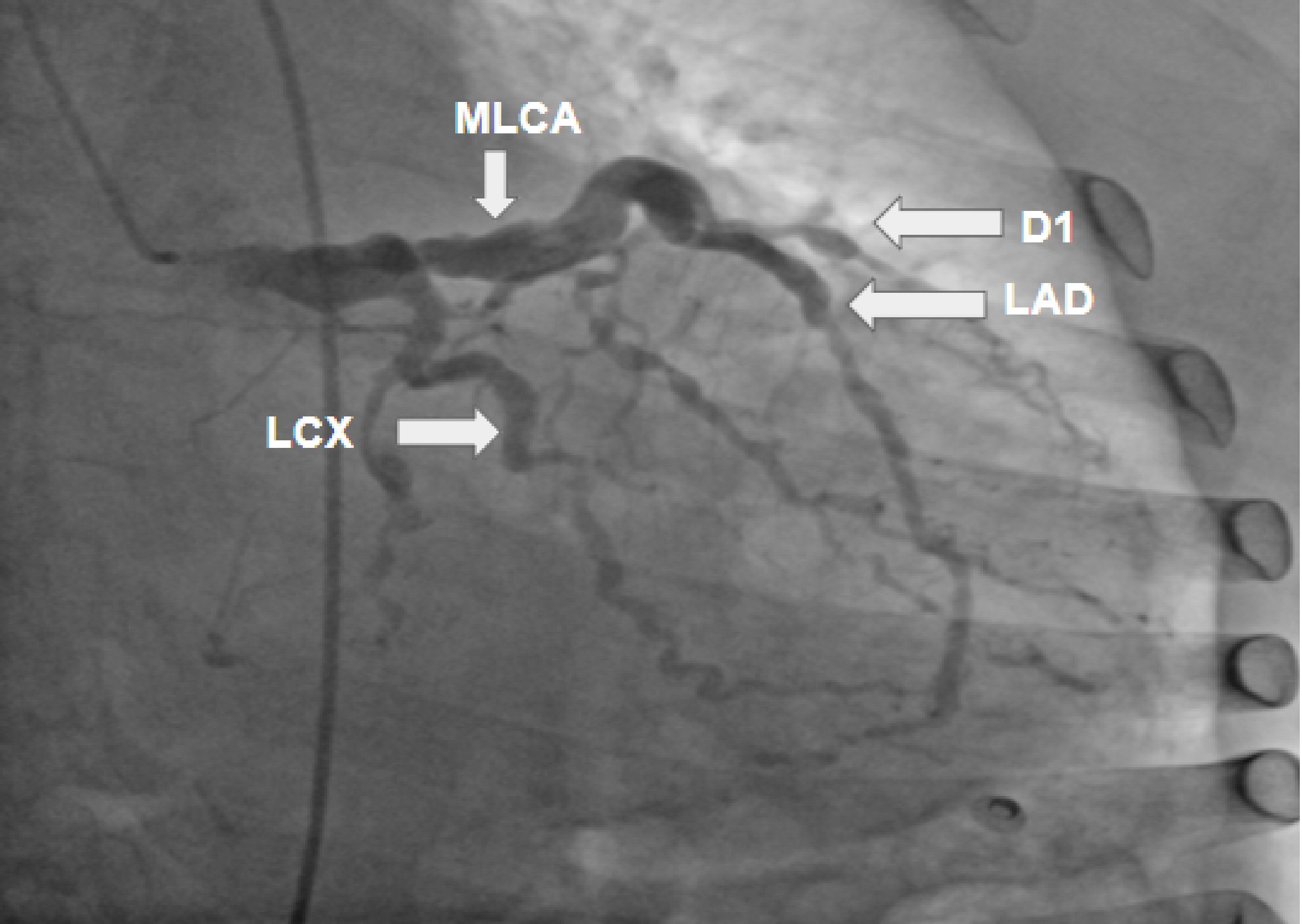 Click for large image | Figure 1. Coronary artery angiogram revealing a severely ectatic left main coronary artery (LMCA); an extremely ectatic left anterior descending (LAD) artery with multiple areas of moderate-to-severe distal and apical stenosis; severely ectatic and stenotic areas of diagonal 1 artery (D1); and multiple areas of stenosis including the first obtuse marginal (OM1) and distal left circumflex (LCX) arteries. |
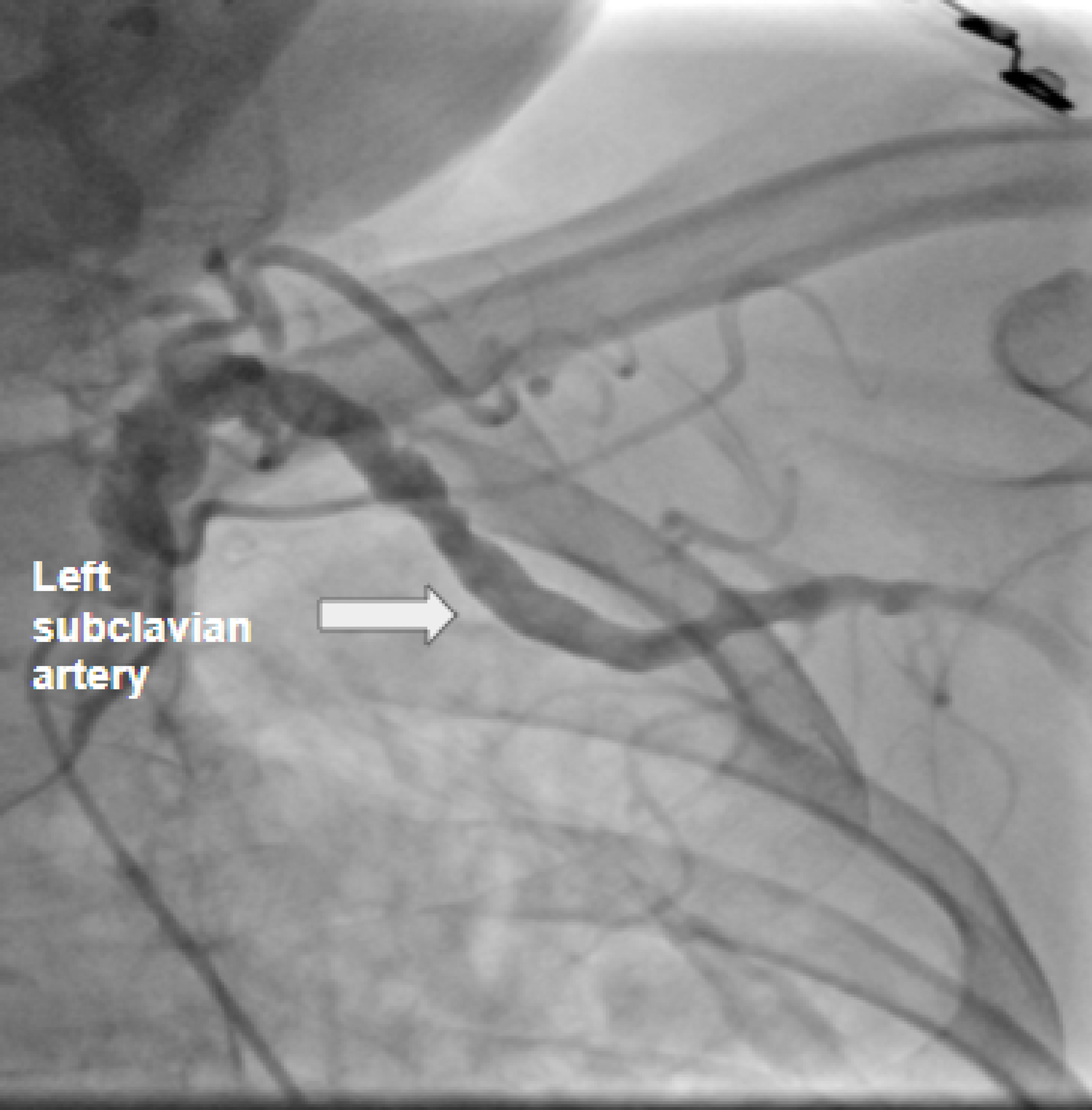 Click for large image | Figure 2. Peripheral coronary angiogram revealing severe ectasia of the left subclavian arteries, with multiple areas of moderate stenosis. |
Moreover, peripheral coronary angiography showed bilateral ectasia of the subclavian arteries, with multiple areas of moderate stenosis. Due to the involvement of numerous arteries, no stent or angioplasty was performed. Ranolazine was started, and the other medications were continued, and the patient was discharged with outpatient follow-up.
Two weeks later, the patient was brought to the hospital on account of dizziness that occurred while doing physical therapy for leg pain. The patient had had multiple episodes of dizziness, but this time she sought medical attention. The patient was alert, oriented for place, person, and times. The neurological exam showed: normal muscle tone; power 5/5; bilateral deep tendon reflex 2+ in the upper and lower limbs; bilateral plantar response; sensory intactness to a light touch; and normal bilateral finger to nose. Orthostatic vitals were negative for orthostatic hypotension. Head computerized tomography (CT) angiography showed: severe IADE involving the vertebrobasilar system, bilateral intracranial internal carotid arteries, and bilateral middle cerebral arteries. There were multiple aneurysms in the left supraclinoid internal carotid artery and bilateral cavernous internal carotid arteries. The sagittal 2D reconstructed images demonstrated a moderate mass effect on the ventral medulla. Considering the severe nature of the IADE and the tortuosity of the vertebrobasilar system, this appearance was likely due to a mass effect on the medulla by the dolichoectatic vertebral-basilar arteries (Fig. 3a-d).
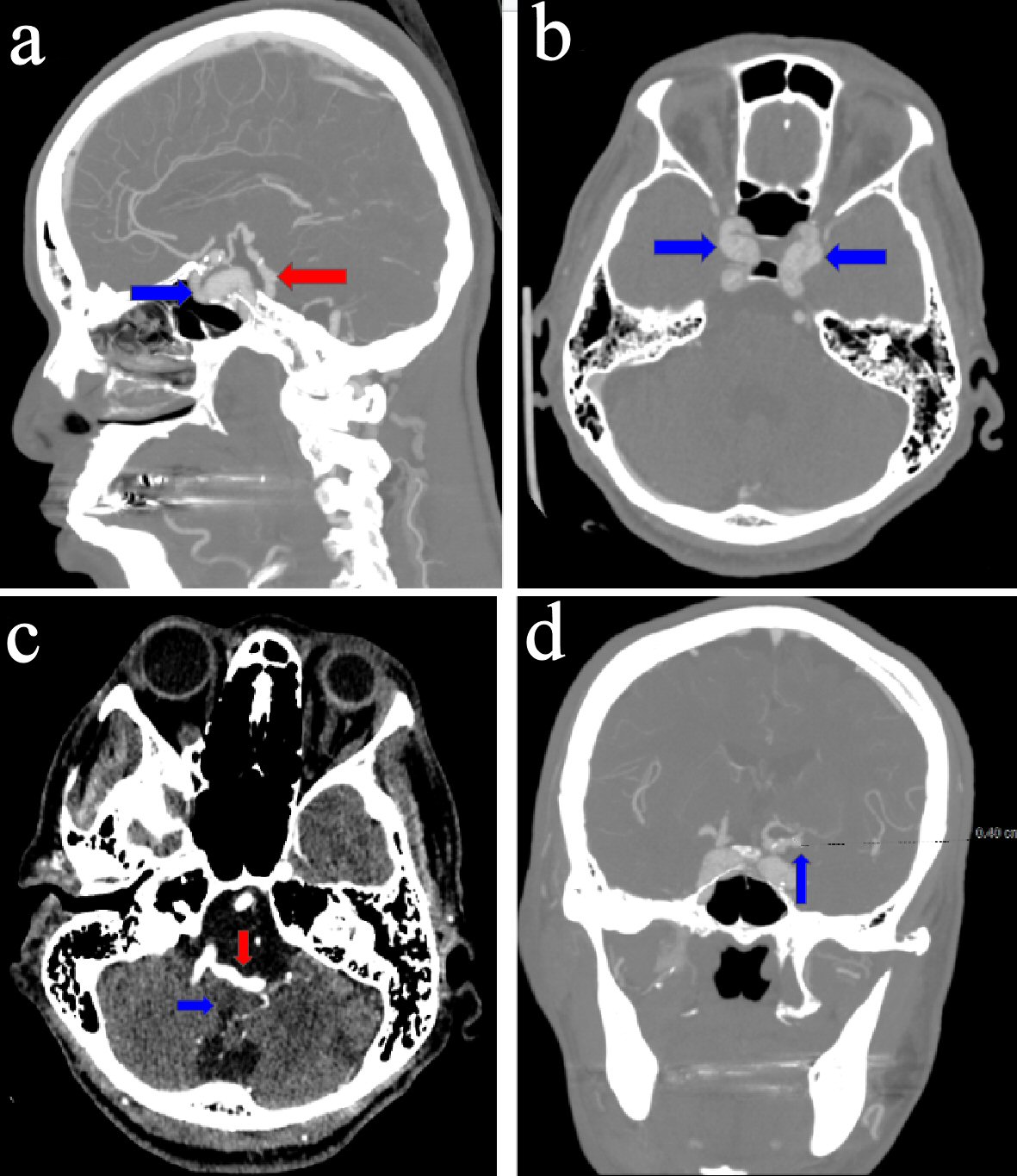 Click for large image | Figure 3. (a) Sagittal image of a computerized tomography (CT) angiogram of the head, revealing ectasia of the vertebrobasilar artery (red arrow) and calcification, dilatation, and ectasia of the intracavernous segment of the left internal carotid artery (blue arrow). (b) Axial image of a CT angiogram of the head, revealing bilateral dilatation and ectasia of the intracavernous segments of the internal carotid arteries (blue arrows). (c) Axial image of a CT angiogram of the head, revealing ectasia involving the vertebrobasilar artery (red arrow) and causing a mass effect on the medulla (blue arrow). (d) Coronal image of a CT angiogram of the head, revealing a 4-mm supraclinoid left internal carotid aneurysm (blue arrow). |
Erythrocyte sedimentation rate, C-reactive protein, chemistry, lipid profile, liver function test, and complete blood count results were within the normal range, except for anemia of iron deficiency, which improved after starting the patient on an iron supplement.
The patient was evaluated by neuro- and neuroendovascular surgical teams, both of which stated that she was not a good candidate for neurovascular intervention due to the complexity and extension of the findings.
The patient was subsequently discharged with outpatient follow-up.
| Discussion | ▴Top |
There exists significant concurrence between CAE and IADE [1]. In one study of more 380 autopsy patients who died of a fatal stroke, 16 had IADE, of which eight had coexistence of CAE [2].
Both conditions are abnormal dilatation of arteries; while CAE affects a coronary arterial segment and leads to a dilatation of at least 1.5 times that of a normal coronary artery [3], IADE affects both the anterior and posterior cerebral circulation, but more frequently involves the intracranial vertebral-basilar arteries [4]. An increased diameter of just one cerebral artery is sufficient to make a diagnosis of IADE [5]. Approximately 20-30% of CAE presentations are congenital, 50% are attributable to atherosclerosis, and 10-20% are associated with inflammatory or connective tissue disease. CAE is diagnosed in approximately 3-8% of patients undergoing coronary angiography, and these patients sometimes present with acute coronary syndrome [3, 6].
On the other hand, IADE is typically found accidentally or when related complications arise. The prevalence is estimated at 0.08-6.5% in the general population; however, patients who have had a cerebrovascular accident have a higher occurrence in the range of 3-17% [4, 5]. Complications of IADE may include ischemic stroke and brain stem compression, which are most commonly seen in vertebrobasilar dolichoectasia with a high 5-year risk. Less frequent complications include subarachnoid hemorrhage and intracerebral bleeding [5, 7-8].
Moreover, risk factors of CAE and IADE have concurrent risk factors including advanced age, male gender, hypertension, and smoking [3, 7]. A history of myocardial infarction has also been documented as a risk factor for vertebrobasilar dolichoectasia [7]. Of note, patients with diabetes mellitus have a low incidence of CAE, which may be due to the downregulation of matrix metalloproteinases (MMPs) with a negative remodeling in response to atherosclerosis. Smoking also appears to be a more common risk factor in patients with CAE than in those with coronary artery disease (CAD), and cocaine use is an independent predictor of CAE, irrespective of smoking [3].
A common pathogenesis has been suggested for CAE and IADE, which implies that dilative arteriopathy may be a systemic process involving several arterial beds simultaneously [2]. This establishes that CAE and IADE are part of a generalized arterial ectasia that is seen in other arteries such as the thoracic and abdominal aorta and the internal carotid arteries [1, 9]. Another pathological pattern of affected muscular arteries has been reported, including rarefaction of the elastic tissue of the media with degeneration of the internal elastic lamina, in addition to MMP-3 dysfunction with the same genotype involvement (5A/5A in the promoter region) [2].
The management of CAE includes drug treatment, angioplasty with a shunt, and surgery. The medical management is contentious, since there is still no sufficient evidence-based approach. Some authors recommend long-term anticoagulation therapy with warfarin based on the significant flow disturbances within ectatic segments [3, 10]; however, this could not be used with our patient, since she had had multiple falls in the past, secondary to dizziness. This therapy has not been tested prospectively and should only be used as a risk vs. benefit approach [3]. It is also suggested that patients (including asymptomatic individuals) be placed on antiplatelet therapy, such as aspirin or adenosine diphosphate (ADP) receptor inhibitors; however, again, this has not yet been evaluated in prospective randomized studies [3]. The role of nitrates or calcium channel blockers for angina remains controversial [11]. With the strong association between CAE and atherosclerosis, guideline-directed medical therapy for risk factors such as hypertension, diabetes, and hyperlipidemia should be considered [6]; medications such as angiotensin-converting enzyme (ACE) inhibitors and statins may be useful in disrupting disease progression [3].
In patients with coexisting obstructive lesions and symptoms or signs of significant ischemia despite medical therapy, percutaneous transluminal coronary angioplasty (PTCA) could be attempted [3]. This technique is relatively challenging, especially with the existence of a large vessel caliber, thrombus burden within the aneurysm, and the potential requirement for a covered stent. Thrombus aspiration can be attempted if identified within the aneurysm. In cases where residual thrombus remains in the artery despite aspiration, some authors advocate for triple therapy (dual antiplatelet treatment and an anticoagulant). Low-dose rivaroxaban (15 mg once daily) has been suggested [12]. There are insufficient data regarding the revascularization of stenotic ectatic coronary arteries. Drug-eluting balloon-expandable stents are used wherever possible. Self-expanding stents (SESs) are advantageous in that they achieve a complete apposition of stent struts as a result of their constant gentle outward force. The newer generation SES STENTYS stent, made from nitinol with nickel-titanium alloy, self-expands, and is confined to the vessel lumen over time [13]. The covered balloon-expandable Jostent has been shown to be capable of treating some coronary aneurysms [14].
In the symptomatic patient who is not suitable for stent insertion, surgical excision, or ligation of the aneurysm with bypass graft of the affected coronary arteries may be the procedure of choice. Coronary artery aneurysm of at least 3 - 4 times the size of the original vessel diameter is considered an absolute indication for surgical intervention by many due to the propensity for complications such as compression, rupture, or thrombosis. Solitary bypass grafting is suggested by some authors as the intervention of choice for non-giant aneurysms [7]. Other surgical options include aneurysm ligation, resection, or marsupialization with interposition grafting [8, 15].
On the other hand, the best protocol for the management of IADE is uncertain. To prevent cerebrovascular accidents, blood pressure control should be prioritized. Antiplatelet therapy may also be indicated for the prevention of recurrent stroke [4]. Patients presenting with compressive neurological manifestations should likely undergo a surgical evaluation [5]. Standard surgical procedures that may be indicated include ventriculoperitoneal shunt insertion for hydrocephalus, microsurgery for trigeminal neuralgia or hemifacial spasms, superficial temporal artery-superior cerebellar artery (STA-SCA) bypass, proximal balloon occlusion, and thrombectomy [7].
Conclusions
CAE and IADE may occur concurrently in a patient since they have similar risk factors and pathogenesis. These are also thought to arise as part of a generalized arterial ectasia, as noticed in our patient; she had ectasia that involved multiple arteries. The management protocol for such a patient is complicated and requires a multidisciplinary team to optimize the patient’s condition. Moreover, there is a need for a more prospective study to identify the best way to treat such a patient.
Acknowledgments
We would like to thank Dr. Richard Grodman in the Cardiology Department and Dr. Joseph Bibawy in the Department of Radiology of our institute for providing us with the test images.
Financial Disclosure
None to declare.
Conflict of Interest
The authors report no conflict of interest.
Informed Consent
Not applicable.
Author Contributions
H. Adnan Alataby is the first author and collected data, guided the literature search, and wrote the manuscript. G. Freg, R. Alzakhari, G. Atteya, and M. Dougar helped with data collection and writing of the article. B. Gala and J. Nfonoyim reviewed and supervised the study.
| References | ▴Top |
- Pico F, Biron Y, Bousser MG, Amarenco P. Concurrent dolichoectasia of basilar and coronary arteries. Neurology. 2005;65(9):1503-1504.
doi pubmed - Pico F, Labreuche J, Hauw JJ, Seilhean D, Duyckaerts C, Amarenco P. Coronary and basilar artery ectasia are associated: results from an autopsy case-control study. Stroke. 2016;47(1):224-227.
doi pubmed - Devabhaktuni S, Mercedes A, Diep J, Ahsan C. Coronary artery ectasia-a review of current literature. Curr Cardiol Rev. 2016;12(4):318-323.
doi pubmed - Kwon HM, Lee YS. Dolichoectasia of the intracranial arteries. Curr Treat Options Cardiovasc Med. 2011;13(3):261-267.
doi pubmed - Del Brutto VJ, Ortiz JG, Biller J. Intracranial arterial dolichoectasia. Front Neurol. 2017;8:344.
doi pubmed - Damay V, Pranata R, Wiharja W. Recurrent acute coronary syndrome in a patient with right coronary artery ectasia: a case report. J Med Case Rep. 2019;13(1):78.
doi pubmed - Wolters FJ, Rinkel GJ, Vergouwen MD. Clinical course and treatment of vertebrobasilar dolichoectasia: a systematic review of the literature. Neurol Res. 2013;35(2):131-137.
doi pubmed - Zdravkovic M, Kostov M, Petrovic S, Zdravkovic J. Dolichoectasia of vertebrobasilar arteries as a cause of hydrocephalus and ischemic cerebral stroke. Am J Forensic Med Pathol. 2017;38(4):359-361.
doi pubmed - Yuh SJ, Alkherayf F, Lesiuk H. Dolichoectasia of the vertebral basilar and internal carotid arteries: A case report and literature review. Surg Neurol Int. 2013;4:153.
doi pubmed - Fukuhara S, Takase E, Fujimoto T, Takami Y, Yamamoto K, Nishida T, Morimoto A. [Coronary ectasia resulting in thrombotic coronary occlusion after warfarin interruption: a case report]. J Cardiol. 2005;46(5):195-200.
- Sorrell VL, Davis MJ, Bove AA. Current knowledge and significance of coronary artery ectasia: a chronologic review of the literature, recommendations for treatment, possible etiologies, and future considerations. Clin Cardiol. 1998;21(3):157-160.
doi pubmed - Korosoglou G, Eisele T, Nunninger P, Giusca S. Mechanical and pharmacologic treatment in a patient with a coronary artery aneurysm and acute coronary syndrome. Coron Artery Dis. 2017;28(4):358-360.
doi pubmed - Moretti C, Omede P, Presutti DG, D'Ascenzo F, De Simone V, Colaci C, Bertaina M, et al. Shaping an ectatic coronary artery: Stentys implantation. Int J Cardiol. 2014;171(3):459-461.
doi pubmed - Szalat A, Durst R, Cohen A, Lotan C. Use of polytetrafluoroethylene-covered stent for treatment of coronary artery aneurysm. Catheter Cardiovasc Interv. 2005;66(2):203-208.
doi pubmed - Beckmann E, Rustum S, Marquardt S, Merz C, Shrestha M, Martens A, Haverich A, et al. Surgical treatment of coronary artery aneurysms. J Card Surg. 2017;32(11):674-679.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


