| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 10, Number 6, June 2019, pages 188-192
Intraoperative Fibrinolysis Associated With Successful Ultralong Resuscitation for Pulmonary Embolism: A Case Report
Pierre Goffina, b, Julien Guntza
aDepartment of Anaesthesia and Intensive Care, CHC de Liege, Belgium
bCorresponding Author: Pierre Goffin, Clinique Saint-Joseph, CHC de Liege, Rue de Hesbaye 75, 4000 Liege, Belgium
Manuscript submitted May 16, 2019, accepted June 21, 2019
Short title: Intraoperative Fibrinolysis
doi: https://doi.org/10.14740/jmc3315
| Abstract | ▴Top |
The present case demonstrates a successful intraoperative administration of fibrinolysis in the setting of cardiac arrest during orthopedic surgery. It was combined with an extralong resuscitation with successful outcome and no complication. No previous case of intraoperative fibrinolysis is reported in the literature and full recovery after extralong resuscitation is only described in a few cases.
Keywords: Pulmonary embolism; Cardiac arrest; Fibrinolysis; Intraoperative; Resuscitation
| Introduction | ▴Top |
Acute thromboembolic events remain a major cause of morbidity and mortality in the perioperative setting. We describe for the first time the successful use of intraoperative fibrinolysis during 125 min of resuscitation after intraoperative cardiac arrest due to pulmonary embolism (PE).
| Case Report | ▴Top |
A 63-year-old man was admitted to our hospital for a scheduled total hip arthroplasty. His medications included antihypertensive therapy and oral antidiabetic. He had no allergy. He benefited from a contralateral hip arthroplasty 2 years earlier without complication. There was no history of thromboembolic event.
The current procedure unfolded without complication. The patient received postoperative thromboprophylaxis with low molecular weight heparin. At day 8 after the surgery, the patient fell and fractured his femur along the prosthesis. Two days after, he was scheduled for open surgical reduction and fixation. This procedure was performed under general anesthesia (GA) and single-shot femoral nerve block with ultrasound guidance.
After 120 min of uncomplicated procedure, the patient became slightly hypotensive and hypoxic, without any response to ephedrine and crystalloid fluid boluses. Within the next 5 min, refractory hypotension and hypocapnia appeared. Cardio-pulmonary resuscitation (CPR) was started with initially a pulseless electrical activity. Despite 25 min of resuscitation efforts (epinephrine, chest compressions with LUCAS® system), a return to spontaneous circulation was not achieved. We monitored our CPR efficacy with end-tidal CO2 measurement about 20 mm Hg and entropy value about 20 (Table 1).
 Click to view | Table 1. Monitoring and Arterial Blood Values |
A bedside echocardiography demonstrated signs of PE. Right ventricular dilatation (indicating acute cor pulmonale) is identified in an apical four-chamber view (Fig. 1). In parasternal short axis view (Fig. 2), images reveal septal flattening (D-shaped left ventricle) consistent with right ventricular pressure overload.
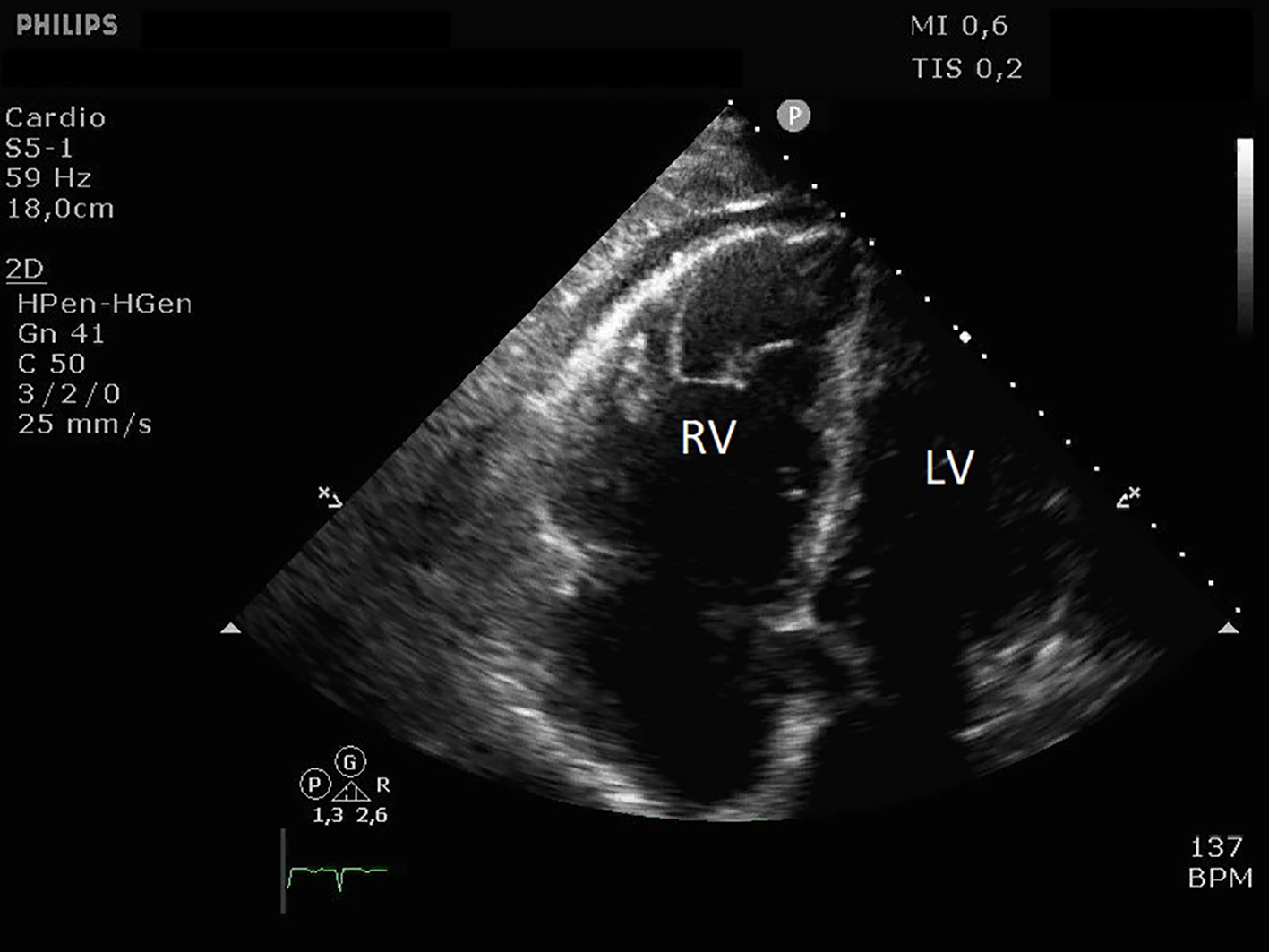 Click for large image | Figure 1. Right ventricular dilatation in apical view transthoracic echocardiography. |
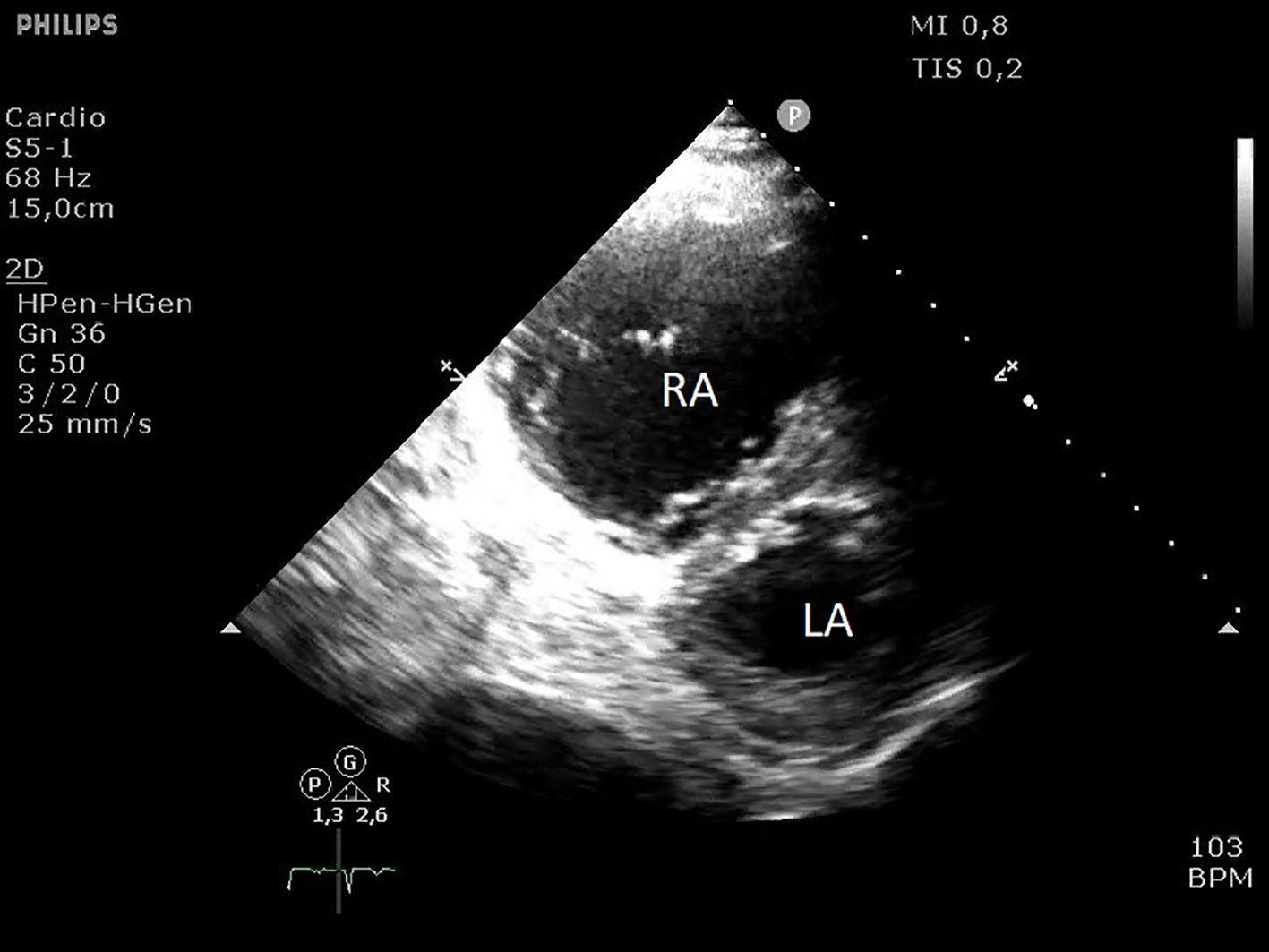 Click for large image | Figure 2. Dilated right atrium and septal flattening in parasternal short axis view transthoracic echocardiography. |
Thirty-five minutes after protocolized CPR, spontaneous circulation was still not re-established. Despite the intraoperative context, we decided to give a fibrinolytic treatment as a last-chance survival option.
While the surgeon summarily closed the surgical site (thigh incision), we inserted a jugular central venous catheter under echography control. Forty-five minutes after the beginning of CPR, we injected the fibrinolytic agent alteplase (10 mg bolus followed by 90 mg into 60 min). We transferred the patient to the intensive care unit (ICU) with the chest compression system ongoing to benefit from a full equipped ICU room. The patient was then started on norepinephrine, dobutamine and received ionic correction (calcium intravenous bolus and bicarbonate administration).
Return of spontaneous circulation was achieved after 80 min in the ICU, with a total CPR time of 125 min. The patient subsequently developed acute kidney injury requiring support with continuous veno-venous hemofiltration. In the following hours, he was stabilized and hemodynamic supportive drugs were slowly weaned off. Two days after, the patient was extubated. He went back later to theater to finish the osteosynthesis. The patient was discharged from ICU at day 32 without neurological deficit.
Six months later, the patient was living independently without focused neurologic deficit. We did not observe any hemorrhagic complication after fibrinolysis.
| Discussion | ▴Top |
Acute PE remains a significant medical problem, accounting for 5-10% of all cardiac arrests and more than 300,000 deaths per year in Europe [1]. The overall mortality is approximately 65% when the patient has a cardiac arrest [2].
The perioperative setting remains a high-risk period for thromboembolic events. The risk of developing a PE is here increased nearly five-fold, leading to an incidence of 0.3-1.6% [3]. Patients undergoing surgery often have underlying risk factors for thrombosis and embolism, such as obesity, smoking, malignancy, or traumatic injury. Furthermore, the inflammatory response to surgery itself leads to a prothrombotic state, which further increases the risk in conjunction with postoperative bed rest and immobilization. Among all surgical specialties, orthopedic surgery represents the major subgroup for thromboembolic risk [3], especially in the case of total hip arthroplasty or hip fracture surgery.
Early detection and treatment are vital for survival. Delayed therapy is identified as an independent predictor of mortality [4]. The perioperative context represents a diagnostic challenge. Signs and symptoms of PE are insidious in a patient under GA which increases the difficulty for early diagnosis. Moreover, the pathophysiologic course of intraoperative PE is affected by procedure and anesthesia. Under GA, the end-tidal CO2 will decrease and the arterial CO2 will increase progressively during the obstruction of pulmonary artery, increasing physiological dead space. Furthermore, low cardiac output and ventilation-perfusion mismatch lead to hypoxemia and hypercapnia [5].
To help the diagnosis, features of acute pulmonary thromboembolism could be identified by transthoracic echocardiography. In case of cardiac arrest, signs are difficult to interpret because right ventricle (RV) is dilated during resuscitation, irrespective of the cause of arrest.
But in studding model of cardiac arrest caused by PE, hypoxia or primary arrhythmia, the RV was more dilated when cardiac arrest was caused by PE [6]. Ultrasonographic findings of RV dilation during cardiac arrest should be interpreted with caution and integrated in the whole clinical context. In this orthopedic perioperative case, the thromboembolic event was extremely probable.
About 30-50% blockade of pulmonary vasculature is necessary to produce hemodynamic instability and shock. Current prognostic stratification strategy defines three situations: massive, submassive and low-risk depending on the presence of shock or sustained hypotension, RV dysfunction or myocardial necrosis [2].
Systemic thrombolysis remains a widely accepted treatment in situation of massive PE. An alternative is catheter-direct thrombolysis technique which allows fragmentation, aspiration and fibrinolytic infusion through a multiperforated catheter spanning the thrombus [2]. Current guidelines acknowledge that it may be considered in patient with massive or submassive PE, especially in high risk bleeding patient or when systemic thrombolysis fails or is contraindicated.
In this context, treatment involves multidisciplinary discussion to assess risk to benefit ratio but the lack of evidence in the perioperative period makes decisions challenging. Although the setting of recent surgery is a relative contraindication for the performance of thrombolysis, it might be a last resort in life-threatening cases with shock, associated with a high risk of major bleeding and hemorrhagic stroke [7].
The European Society of Cardiology recommends considering the use of fibrinolytic therapy as the first-line treatment when cardiac arrest is caused by proven or suspected acute PE [8]. Given the bleeding risk associated with thrombolytic therapy, the treatment should only be administrated after balancing the potential benefits of improved outcomes and bleeding risk of thrombolytic therapy.
Among patients with a high risk of PE, thrombolytic therapy is associated with a lower rate of all-cause mortality but an increased risk of major bleeding. Bleeding was reported to occur more or less in 13% of patients receiving thrombolytic therapy including 1.5% of intracranial hemorrhages [7]. But there are no data in case of perioperative thrombolysis.
The same review confirms this significant reduction in mortality and reminds us that recent surgery or trauma within 10 days is a relative contraindication. However, these relative contraindications are also associated with inducible risks for PE. Therefore, thrombolytic therapy may still be appropriate for patients with massive PE complicated by relative contraindications.
We decided to perform a thrombolytic treatment after balancing the high risk of surgical bleeding and the fatal issue of a failing resuscitation. We used a central jugular venous catheter to allow central injection of thrombolysis. It is thought that central perfusion may be more effective than peripheral infusion by increasing concentration of tissue plasminogen activator at the site of the thrombus [9]. We did not observe any haemorrhagic complication at surgical site nor at the puncture site of femoral nerve block.
In this case, immediate surgical or endovascular therapy was not feasible due to patient conditions. Extracorporeal membrane oxygenation (ECMO) is unavailable in our institution. The jugular directed dose of thrombolytics in conjunction with CPR was associated with the survival of the patient.
Long resuscitation
It appears that long resuscitation is associated with worse outcome [10]. In his observational study of CPR, Youness et al [11] have shown a median survival rate of 25.5% descending to 5.6% when duration of CPR was more than 30 min.
Current literature suggests a negative correlation between length of resuscitation and mortality. There is no guideline for ideal resuscitation time. In a large registry of 64,339 patients [11], the median duration of resuscitation was 12 min (interquartile range (IQR): 6 - 21 min) in the 15.4% of survivors compared with 20 min (IQR: 14 - 30 min) for all other non-survivors.
It is really difficult to define an optimum duration for resuscitation but some signs can indicate that CPR is performed well. We observed in our CPR, an increase in end-tidal CO2 and entropy value after the initial collapse. These values remained stable during resuscitation, probably indicating an acceptable cerebral flow and oxygenation. Depth of anesthesia monitoring during CPR has also been used by some authors to assess cerebral perfusion as well as adequacy of chest compression [11] (Table 1).
Only few cases of ultralong resuscitation have been published [11]. In our case report, we performed a CPR of 125 min with excellent outcome. The same authors conclude that decision to continue or stop CPR should not be based solely on the duration of resuscitation. It must include the cause of the arrest, the latency to starting CPR, the quality of CPR and morbidity of the patient, factors that affect the outcome. When potentially reversible cause of cardiac arrest has been diagnosed, it usually admitted to continue CPR until the reversible cause is treated.
Conclusion
In this case, we performed a successful ultralong resuscitation as rarely described in the literature, combined with the first intraoperative fibrinolysis without hemorrhagic complication. This case emphasizes the importance of following a high-quality resuscitation protocol. It also reminds us to balance the risk-to-benefit ratio in case of an emergency fibrinolysis, especially in the perioperative settings.
Acknowledgments
This case was presented at ESRA Congress in September 2018 in Dublin (Ireland).
Financial Disclosure
None.
Conflict of Interest
None.
Informed Consent
Written consent to publish this case was obtained from the patient.
Author Contributions
Pierre Goffin is the main author. Julien Guntz helped the manuscript revision.
Abbreviations
PE: pulmonary embolism; CPR: cardio-pulmonary resuscitation; ICU: intensive care unit; GA: general anesthesia; RV: right ventricle
| References | ▴Top |
- Gordon RJ, Lombard FW. Perioperative venous thromboembolism: a review. Anesth Analg. 2017;125(2):403-412.
doi pubmed - Ruohoniemi DM, Sista AK, Doany CF, Heerdt PM. Perioperative pulmonary thromboembolism: current concepts and treatment options. Curr Opin Anaesthesiol. 2018;31(1):75-82.
doi pubmed - Desciak MC, Martin DE. Perioperative pulmonary embolism: diagnosis and anesthetic management. J Clin Anesth. 2011;23(2):153-165.
doi pubmed - Beydilli I, Yilmaz F, Sonmez BM, Kozaci N, Yilmaz A, Toksul IH, Guven R, et al. Thrombolytic therapy delay is independent predictor of mortality in acute pulmonary embolism at emergency service. Kaohsiung J Med Sci. 2016;32(11):572-578.
doi pubmed - Mao Y, Wen S, Chen G, Zhang W, Ai Y, Yuan J. Management of intra-operative acute pulmonary embolism during general anesthesia: a case report. BMC Anesthesiol. 2017;17(1):67.
doi pubmed - Granfeldt A, Lofgren B. Detection of pulmonary embolism during cardiac arrest -ultrasonographic findings should be interpreted with caution. CCM. 2017;(3):1-8.
- Yamamoto T. Management of patients with high-risk pulmonary embolism: a narrative review. J Intensive Care. 2018;6:16.
doi pubmed - Konstantinides SV, Torbicki A, Agnelli G, Danchin N, Fitzmaurice D, Galie N, Gibbs JS, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2014;35(43):3033-3069, 3069a-3069k.
- Gulati V, Brazg J. Central Venous Catheter-directed Tissue Plasminogen Activator in Massive Pulmonary Embolism. Clin Pract Cases Emerg Med. 2018;2(1):67-70.
doi pubmed - Reynolds JC, Frisch A, Rittenberger JC, Callaway CW. Duration of resuscitation efforts and functional outcome after out-of-hospital cardiac arrest: when should we change to novel therapies? Circulation. 2013;128(23):2488-2494.
doi pubmed - Youness H, Al Halabi T, Hussein H, Awab A, Jones K, Keddissi J. Review and outcome of prolonged cardiopulmonary resuscitation. Crit Care Res Pract. 2016;2016:7384649.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


