| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 5, Number 7, July 2014, pages 420-422
Drug Rash With Eosinophilia and Systemic Symptoms Syndrome Induced by Allopurınol
Mukaddes Kavalaa, Ayse Serap Karadaga, Filiz Topaloglu Demira, c, Ilkin Zindancia, Zafer Turkoglua, Berkant Omana, Ebru Zemherib, Emin Ozlua
aDepartment of Dermatology, S.B Istanbul Medeniyet University, Goztepe Training and Research Hospital, Istanbul, Turkey
bDepartment of Pathology, S.B Istanbul Medeniyet University, Goztepe Training and Research Hospital, Istanbul, Turkey
cCorresponding Author: Filiz Topaloglu Demir, Department of Dermatology, S.B Istanbul Medeniyet University, Goztepe Training and Research Hospital, Istanbul, Turkey
Manuscript accepted for publication May 19, 2014
Short title: DRESS Syndrome
doi: https://doi.org/10.14740/jmc1811w
| Abstract | ▴Top |
Drug rash with eosinophilia and systemic symptoms (DRESS) syndrome is an adverse drug reaction caused by an apparent group of drugs which can cause 10-20% mortality. It is characterized by a latency period ranging from 3 weeks to 3 months after the introduction of the offending drug. The syndrome is defined by the presence of fever, rash, eosinophilia, atypical lymphocytes and multiorgan involvement. We present a 39-year-old woman who developed fever, nausea, a pruritic erythematous maculopapular rash and facial edema during her sixth week of the treatment with allopurinol as a case of DRESS syndrome. Diagnosis was confirmed by the drug rash, eosinophilia and systemic involvement including adenopathy, toxic hepatitis and pericardial effusion. Allopurinol was discontinued and intravenous prednisolone 60 mg/day was started. The patient’s clinical appearance and eosinophilia improved within first 2 days. Awareness of hematologic abnormalities and systemic involvement along with drug rush, by physicians is critical for early diagnosis of this life-threatening syndrome.
Keywords: DRESS syndrome; Allopurinol; Drug reactions
| Introduction | ▴Top |
Drug rash with eosinophilia and systemic symptoms (DRESS) syndrome, previously named “drug hypersensitivity syndrome” is severe adverse drug-induced reaction. Diagnosis of DRESS syndrome is challenging, because the pattern of cutaneous eruption and the types of organs involved are various. We describe a case of DRESS syndrome induced by allopurinol, and emphasize the importance of heterogeneity of clinical presentation which often results in initial misdiagnosis.
| Case Report | ▴Top |
A 39-year-old woman with a history of fever, malaise, pharyngitis and diffuse maculopapular rash on her body was admitted to the infection diseases department. She was started on amoxicillin/clavulanic acid treatment for a suspected upper respiratory tract infection. Five days after starting the previously mentioned antibiotic, she was consulted to us and transferred to our dermatology department. Her medical history was notable for appendectomy and pituitary gland tumor operation 3 and 5 years ago, and she had been treated with allopurinol for increased uric acid levels. Seven weeks after the initiation of allopurinol she had developed a skin eruption which began on the trunk and upper limbs and gradually disseminated for a couple of days. Physical examination revealed a generalized maculopapular exanthema, some of them with edematous follicular accentuation, periorbital edema and generalized lymphadenopathy (Fig. 1a, b). The patient was febrile (38.2 °C).
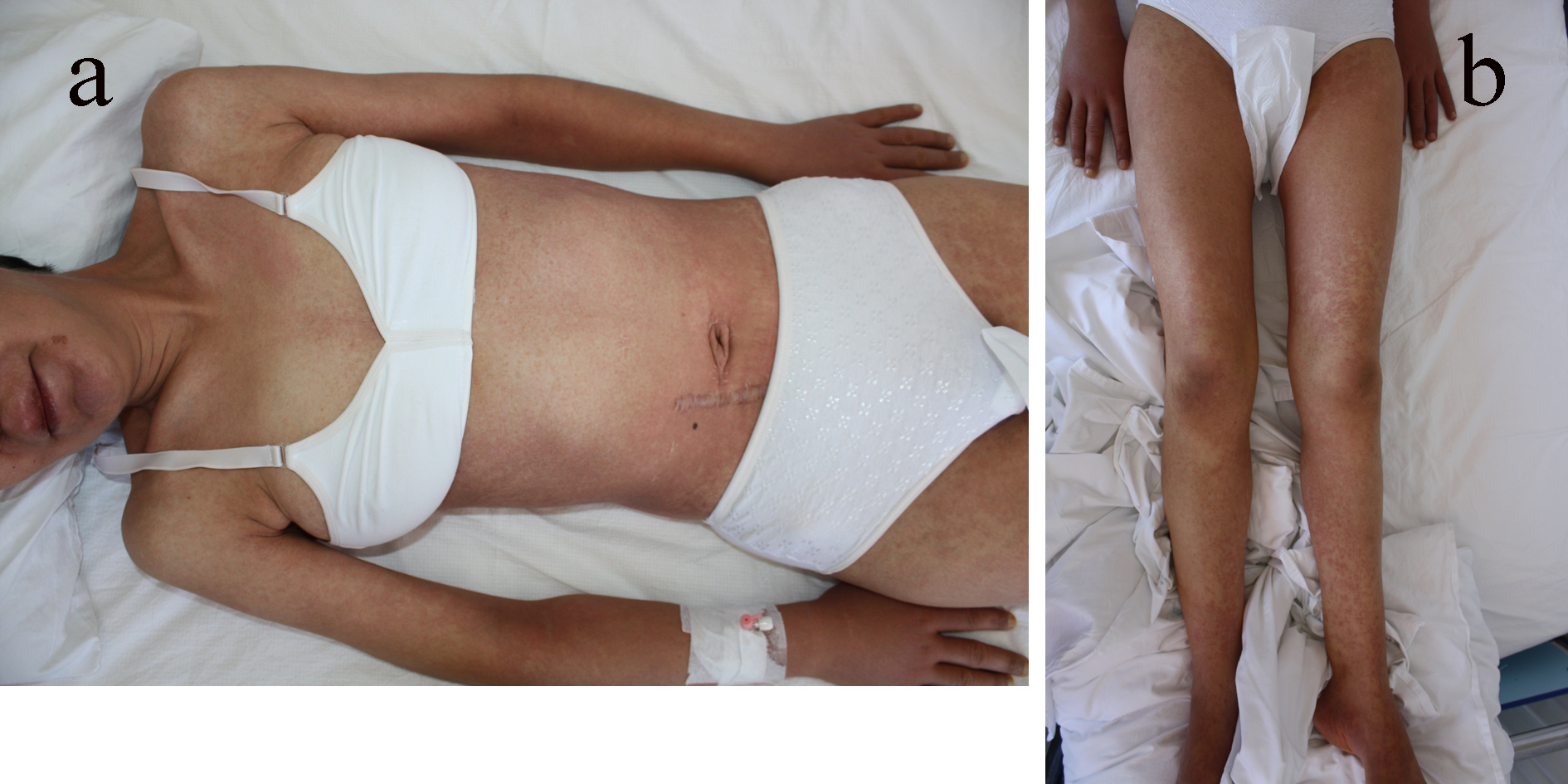 Click for large image | Figure 1. (a) A generalized maculopapular exanthema on the trunk and upper extremities. (b) A maculopapular exanthema on lower extremities. |
Laboratory findings showed a WBC count of 14 × 109/L with 7.6% eosinophils. Liver function tests were abnormal with increased levels of AST (455 U/L, N: 0 - 35 U/L), ALT (565 U/L, N: 0 - 35 U/L), GGT (532 U/L, N: 0 - 50 U/L) and alkaline phophatase (346 IU/L, N: 30 - 120 U/L). There was an increase in serum creatinine (1.64 mg/dL, N: 0.50 - 9 mg/dL) and urea (89 mg/dL, N: 17 - 43 mg/dL) and decreased creatinine clearance (61.72 mL/min, N: 85 - 125 mL/min). Blood cultures, urine and stool cultures and nasal-throat swabs were negative for bacteria, protozoa or helminthic eggs. Serologic tests including syphilis, EBV, HIV and hepatitis B and C and antinuclear antibody were negative. Chest radiograhy was normal. Echocardiography showed pericardial effusion 0.8 cm around left ventricle and abdominal sonography showed moderate hepatomegaly. The skin biopsy yielded mild acanthosis and leukocytoclastic vasculitis in the upper dermis (Fig. 2). Allopurinol was immediately withdrawn, and systemic prednisolone 75 mg daily and antihistamines were initiated. The patient’s clinical appearance and laboratory abnormalities began to improve. The dosage of corticosteroids was gradually tapered and stopped as the clinical and laboratory abnormalities resolved over 6 months in order to prevent a recurrence of the symptoms of the disease.
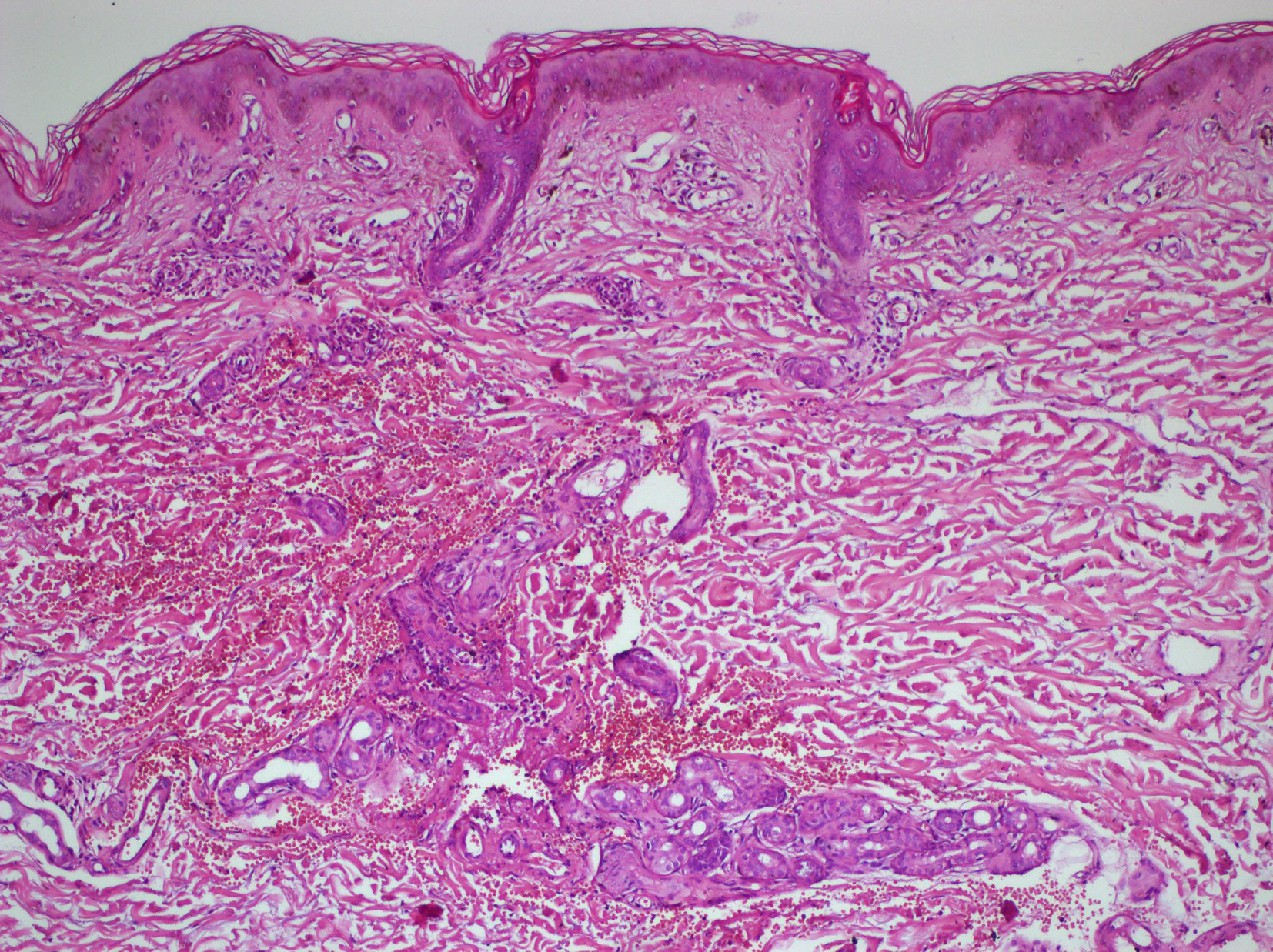 Click for large image | Figure 2. H&E (× 20). Mild acanthosis and leucocythoclastic vasculitis in the upper dermis. |
| Discussion | ▴Top |
DRESS syndrome is a potentially life-threatening syndrome including a severe skin eruption, fever, hematologic abnormalities and internal organ involvement. The mortality rate has been estimated at 10% [1, 2]. DRESS syndrome most commonly arises 3 - 8 weeks after exposure to the culprit drug. Our patient presented to the infection disease department 6 weeks after initiation of allopurinol. Fever often up to 38 - 40 °C and rash is the first sign seen in 90-100% and 90% of cases respectively [3]. The face, trunk and upper extremities are initially affected and the eruption is usually macular erythema that becomes confluent. Swelling of the face, with a marked periorbital involvement is a warning to the diagnosis, occurring in about 25% of patients [4]. Local or generalized lymphadenopathy is found in 70-75% of cases [5]. Various hematologic abnormalities are observed including leukocytosis, eosinophilia and atypical lymphocytes similar to mononucleosis. Liver involvement is the most common visceral manifestation, after the lymphadenopathy. Hepatitis with elevation of liver transaminases is found in 51% of cases [2]. Renal involvement is also reported in patients, being particularly evident in cases due to allopurinol [6]. Our patient’s constellation of symptoms and course were consistent with the natural progression of DRESS syndrome. Severe organ involvement may develop, such as fulminant hepatitis, eosinophilic interstitial lung disease, interstitial nephropathy, pericarditis, myocarditis and pancreatitis, 1 - 2 weeks after skin eruption [1, 2].
The aromatic anticonvulsants, salazosulfapyridine, dapsone and minocycline are frequently associated with DRESS syndrome, but a variety of other drugs can induce this syndrome [1]. Although the pathogenesis is unclear, different mechanisms have been implicated involving detoxification defects, immunological reactions, slow acetylation and reactivation of human herpes virus 6 [5-8].
Therapeutic approach consists of immediate discontinuation of the offending drug and administration of systemic corticosteroids, as the first choice. Intravenous immunoglobulins, plasmapheresis or a combination of these therapies can also be used if the symptoms get worse [4].
A correct and early diagnosis is necessary as DRESS syndrome is potentially associated with serious and sometimes fatal visceral involvement.
Conflict of Interest
There are no conflicts of interest among all authors regarding this article.
| References | ▴Top |
- Tas S, Simonart T. Management of drug rash with eosinophilia and systemic symptoms (DRESS syndrome): an update. Dermatology. 2003;206(4):353-356.
doi - Michel F, Navellou JC, Ferraud D, Toussirot E, Wendling D. DRESS syndrome in a patient on sulfasalazine for rheumatoid arthritis. Joint Bone Spine. 2005;72(1):82-85.
doi pubmed - Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (Drug Rash with Eosinophilia and Systemic Symptoms: DRESS). Semin Cutan Med Surg. 1996;15(4):250-257.
doi - Shear NH, Spielberg SP. Anticonvulsant hypersensitivity syndrome. In vitro assessment of risk. J Clin Invest. 1988;82(6):1826-1832.
doi pubmed - Descamps V, Valance A, Edlinger C, Fillet AM, Grossin M, Lebrun-Vignes B, Belaich S,
et al . Association of human herpesvirus 6 infection with drug reaction with eosinophilia and systemic symptoms. Arch Dermatol. 2001;137(3):301-304.
pubmed - Picard D, Janela B, Descamps V, D'Incan M, Courville P, Jacquot S, Rogez S,
et al . Drug reaction with eosinophilia and systemic symptoms (DRESS): a multiorgan antiviral T cell response. Sci Transl Med. 2010;2(46):46-62.
doi pubmed - Shiohara T, Inaoka M, Kano Y. Drug-induced hypersensitivity syndrome (DIHS): a reaction induced by a complex interplay among herpesviruses and antiviral and antidrug immune responses. Allergol Int. 2006;55(1):1-8.
doi pubmed - Kito Y, Ito T, Tokura Y, Hashizume H. High-dose intravenous immunoglobulin monotherapy for drug-induced hypersensitivity syndrome. Acta Derm Venereol. 2012;92(1):100-101.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


