| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 5, Number 6, June 2014, pages 329-333
Magnetic Resonance Imaging in Addition to Ultrasound in the Diagnosis of Cesarean Scar Ectopic Pregnancy
Duygu Kara Bozkurta, Murat Bozkurtb, d, Levent Sahinc
aDepartment of Radiology, Kafkas University School of Medicine, Kars, Turkey
bDepartment of Gynecology and Obstetric, Kafkas University School of Medicine, Kars, Turkey
cPark Hospital IVF Center, Malatya, Turkey
dCorresponding author: Murat Bozkurt, Kafkas Universitesi Saglik Arastırma ve Uygulama Hastanesi Kars, Turkey
Manuscript accepted for publication March 27, 2014
Short title: Magnetic Resonance Imaging
doi: https://doi.org/10.14740/jmc1734w
| Abstract | ▴Top |
Ectopic pregnancies rising from a cesarean scar are very rare. The diagnosis rate and the incidence of cesarean scar pregnancy have been increasing because of the increasing number of births by cesarean section and development in transvaginal ultrasound (TVUS) and color Doppler ultrasonography. Early diagnosis is very important in cesarean section scar pregnancies because of complications such as massive bleeding and uterine rupture that may cause maternal mortality. TVUS is the first method to be used for diagnosis in routine practice. Magnetic resonance imaging (MRI) is also used in addition to transvaginal ultrasonography in some cases. In this case report patient was suspected to have cervical ectopic pregnancy after TVUS examination; however, it was understood that the diagnosis was found to be compatible with cesarean scar pregnancy after MRI. This situation demonstrates that MRI can be used in addition to TVUS in some patients who cannot be diagnosed easily by transvaginal ultrasonography. In this case report we discussed the diagnosis and the treatment of cesarean scar pregnancy with recent literature, and the importance of using MRI was emphasized in difficult cases.
Keywords: Ectopic pregnancy; Cesarean scar pregnancy; Magnetic resonance imaging; Transvaginal ultrasonography
| Introduction | ▴Top |
Cesarean scar ectopic pregnancy is extremely rare but its incidence has been increasing because of the increasing rate of births by cesarean section. The incidence of cesarean scar ectopic pregnancy from all pregnancies is 0.15% (1:2,226). The incidence of cesarean scar pregnancy from all ectopic pregnancy is 6.1% [1]. Gestational age ranges from 5 weeks to 12 weeks and 4 days at diagnosis [2]. Early diagnosis is important because maternal mortality increases with increasing gestational weeks. Cervical pregnancy, incomplete abortion and cervico-isthmic pregnancy should be considered for the differential diagnosis of cesarean scar pregnancy. The transvaginal ultrasonography is the most commonly used imaging modality for differential diagnosis. Sonographic criteria, which could be helpful in diagnosis of cesarean scar pregnancy, are defined by Vial et al; however, transvaginal ultrasonography cannot put a definitive diagnosis in all cases [3]. In this case report, a cesarean section scar pregnancy cannot be ruled out in patients who suspected of having cervical pregnancy by TVUS and clinical findings. For this reason MRI was performed additional to TVUS. After MRI, the diagnosis was changed to post-cesarean scar pregnancy. In this presentation, the clinical and radiological diagnosis of cesarean scar pregnancy, the advantages of MRI and treatment options were discussed with current literature.
| Case Report | ▴Top |
HB, a 39-year-old woman gravida 4, para 2 was referred to Kafkas University School of Medicine, Gynecology and Obstetric Department, approximately 7 weeks after her last normal menstrual period (LMP). She had a mild abdominal pain and a vaginal bleeding for 2 days. She had a history of two prior cesarean deliveries, 4 and 2 years ago, and a curettage following a spontaneous abortion. On vaginal examination, the uterus was mild tender, external cervical os was closed and clots with minor bleeding was seen. Blood pressure was 100/60 mm Hg and heart rate was 92 beats per minute (BPM) on physical examination. She was hemodynamically stable. Her laboratory examination showed that hemoglobin level was 12.1 g/dL, platelet count was 226 × 103/µL and white blood cell count was 12.6 × 103/µL (3.5 - 10.5). Prothrombin time was 13.9 s (10 - 15 s), and INR was 1.06 (0.8 - 1.2). The β-human chorionic gonadotrophin (β-hCG) level (13.405 mIU/mL) was significantly elevated. Liver and kidney function tests were in normal limits. Electrolyte imbalance was not observed. TVUS was performed by using VOLUSON (GE Healthcare Technologies, Ultrasound, Milwaukee, USA) transvaginal (7.5 MHz) transducer. The ultrasound examination revealed that there was a retrovert, normal uterine fundus with a thick endometrium. The uterine cavity was empty. There was a gestational sac at the level of the lower uterine segment. The crown-rump length was 4.9 mm which corresponds to an estimated gestational age of 6 weeks and 1 day (Fig. 1). A possible uterine rupture was ruled out because there was no free fluid in Douglas. The fetal heart rate was seen in color Doppler ultrasound examination but it was bradycardic (50 - 60 BPM). A peak velocity of 35 cm/s and pulsatility index of 0.8 were measured in pulse Doppler examination. A localized cervical ectopic pregnancy was considered in the first place. However, due to the patient’s history of two previous cesarean sections, possible cesarean scar pregnancy could not be ruled out. Pelvic MRI was performed in a 1.5 Tesla MR unit (Magnetom Avanto, Siemens, USA) in order to localize the lesion more clearly. Sagittal T2 turbo spin echo (TSE), fat-suppressed sagittal, axial and coronal T2-TSE sequences were performed. A gestational sac which protrudes from myometrium at the anterior wall of the lower uterine segment was monitored. A very thin myometrium in the central part of the sac and thin serosa layer was viewed around the sac. Bladder wall integrity was preserved and the wall invasion was not detected. Uterine and cervical cavity was empty (Fig. 2). As a result of MRI findings, patient was diagnosed in cesarean scar ectopic pregnancy without bladder invasion. Systemic methotrexate therapy (1 mg/kg) was initiated. In the first day, β-hCG level was not changed, fetal heart rate was observed and vaginal bleeding was persisted. The second dose intramuscular methotrexate therapy (1 mg/kg) was administered 4 days later but the serum β-hCG levels were decreased minimally; follow-up ultrasound examinations show that fetal cardiac activity and vaginal bleeding continued. Therefore, D & C was done. Patient’s ultrasound controls were normal after therapy, and β-hCG level reached normal levels at sixth week.
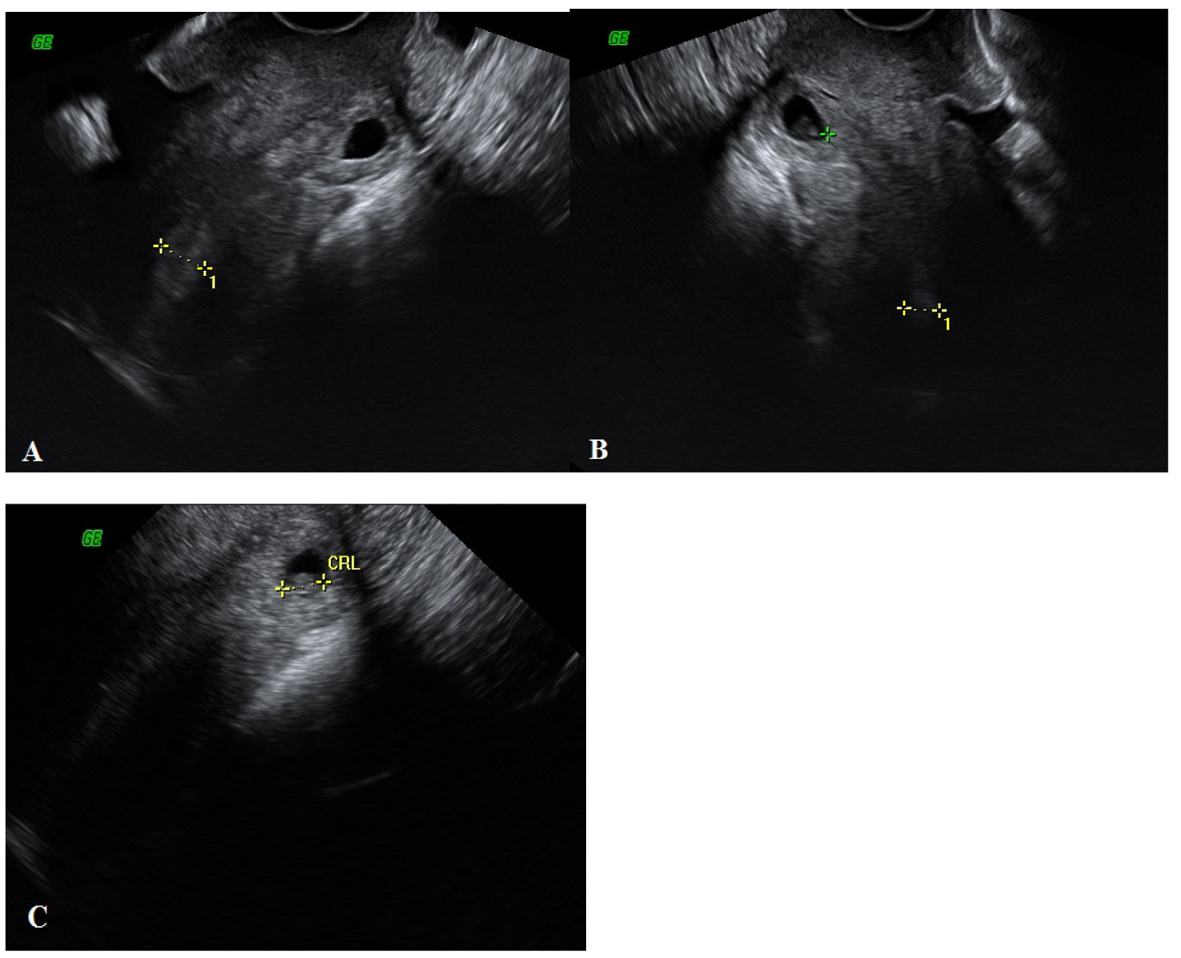 Click for large image | Figure 1. (A, B, C) At the level of the cervical canal there was an ectopic gestational sac including crown-rump length measuring 7.6 mm corresponding to an estimated gestational age of 6 weeks and 5 days. |
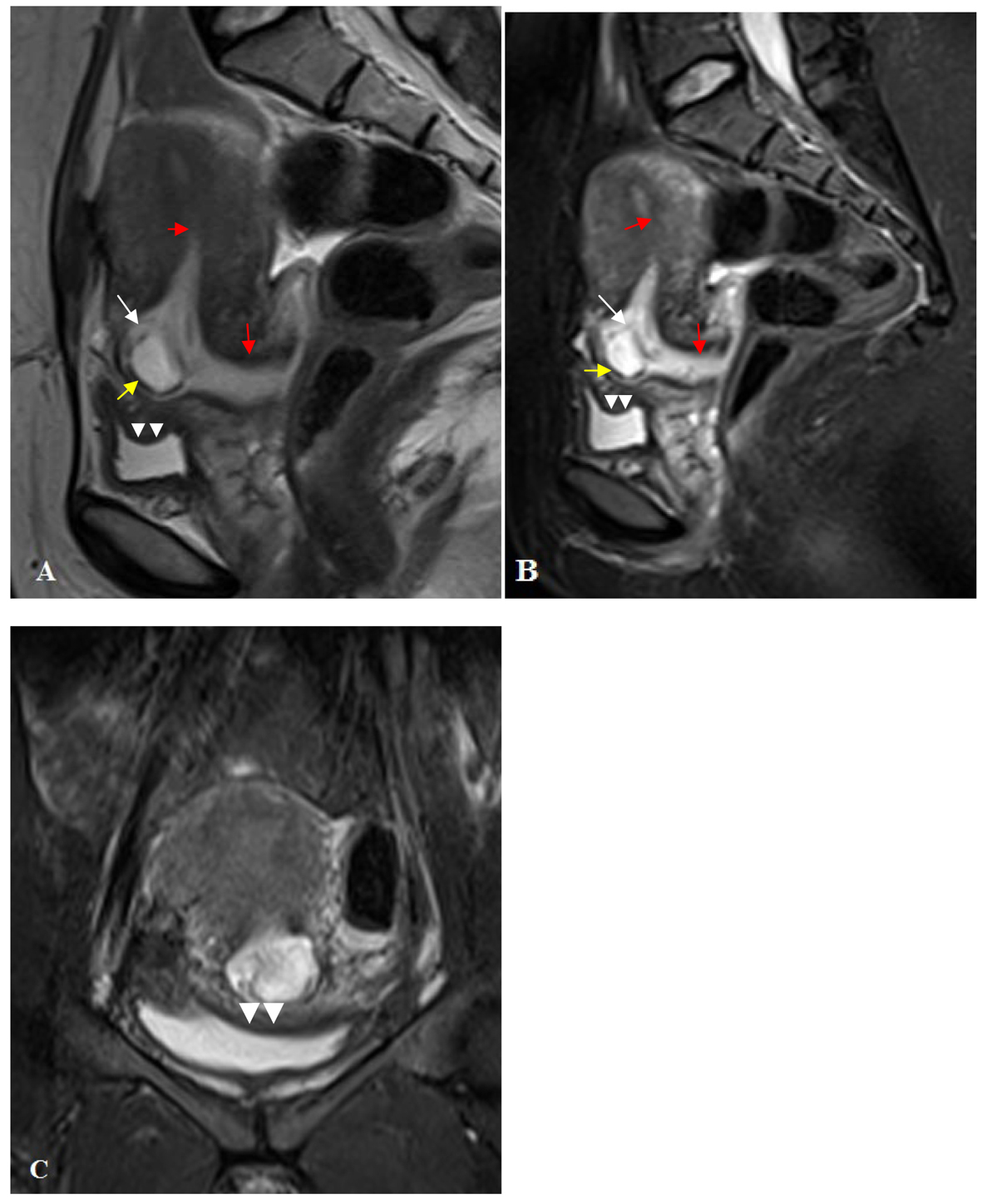 Click for large image | Figure 2. (A) Sagittal plan T2 TSE sequence; (B) Sagittal plan T2 FS TSE; (C) Coronal plan T2 FS TSE sequences shows a gestational sac which protrudes from myometrium at the anterior wall of the lower uterine segment (white arrow). A very thin myometrium in the central part of the gestational sac and thin serosa layer was viewed around the sac (yellow arrow). Bladder wall integrity was preserved and the wall invasion was not detected (arrow head). Uterine and cervical cavity was empty (red arrows). |
| Discussion | ▴Top |
The pathophysiology and etiology of cesarean scar pregnancy has not been fully understood and different theories have been proposed in the literature. According to one hypothesis, a poor vascularity in the anterior lower uterine segment disrupts healing process after cesarean procedures in some women; therefore, this area is vulnerable to form small dehiscent tracts or defects where a trophoblast can implant [1]. Most define the embryo entering the myometrium through microscopic tracts from small uterine dehiscences, with the lack of decidua basalis over the scar. This would explain why the gestational sac in a cesarean scar pregnancy is completely embedded in the myometrium, surrounded by fibrous scar tissue and separate from the endometrial cavity [4]. In women who underwent cesarean section more than one is at increased risk of scar implantation caused by the formation large scar areas. This situation is observed not only following cesarean section but also after curettage, myomectomy, metroplasty, hysteroscopy and manual removal of the placenta [1]. In some studies, there was no correlation between the number of previous cesarean section and the number of scar tissue ectopic pregnancies [5, 6]. On the other hand, other studies demonstrated that especially patients who had three or more cesarean sections have an increased risk of scar tissue ectopic pregnancy but patients undergoing operation once or twice have no significantly increased risks of ectopic scar pregnancy [7, 8]. In our case, the patient had a history of two cesarean sections and D & C.
Patient’s medical history supports the idea that the uterine wall damage may be associated with cesarean scar pregnancy. Medical history and physical examination findings are very important in the diagnosis of cesarean scar pregnancy as in other obstetric and gynecologic diseases. Seow et al indicated that the amount of vaginal bleeding and abdominal pain pattern could provide important clues in the differential diagnosis of cesarean scar pregnancy. They reported that massive vaginal bleeding and cramp style severe abdominal pain occur in spontaneous abortion secondary to detachment of chorionic sac; on the other hand, mild vaginal bleeding and moderate abdominal pain may occur in scar pregnancy [6]. In our case, there were slight vaginal bleeding and abdominal tenderness continuing for 2 days.
TVUS should be the first imaging modality to be used for diagnosis after medical history and physical examination findings. The ectopic pregnancy can be diagnosed by ultrasound in most of the cases. Sonographic criteria which may be helpful in diagnosis of cesarean scar pregnancy were defined by Vial et al: 1) a trophoblast located between the bladder and anterior uterine wall at the presumed site of the cesarean section scar; 2) a gestational sac which is ovoid and regular in shape, as opposed to distorted and collapsed as can be seen in miscarriages and 3) a thin or discontinuous myometrium between the gestational sac and urinary bladder wall on sagittal images of the uterus through the amniotic sac [3]. Vascularity of the sac on color Doppler interrogation can aid in distinguishing a CSP from the avascular sac of an aborting pregnancy. Alternatively, a negative “sliding organ sign” when gentle pressure is applied to a sac seen at the level of the internal orifice of the uterus using the endovaginal probe can help differentiate a CSP from a spontaneous abortion in progress. Findings of high velocity (peak velocity > 20 cm/s) and low impedance (pulsatility index < 1) waveforms on pulsed Doppler have been described with scar implantation [9].
Cesarean section scar pregnancies are often diagnosed by transvaginal ultrasonography and Doppler findings but if there is a difficulty in diagnosing cases as in our patient, MRI can be used. MRI is very helpful in showing sac characterization, localization and whether there is an invasion of adjacent organs because it is a multiplanar imaging method and it has a high soft tissue resolution. Another advantage of the MRI is that it gives critical information for further treatment plan and guidance to surgeons [10].
MRI’s disadvantages are the high cost and long duration of shooting. Patients with massive bleeding should be monitored during MRI shooting and the length of exposure time should be kept in mind.
In our patient, the ultrasound findings showed that there was an ectopic pregnancy at the lower uterine segment, but we could not distinguish whether it was a cervical pregnancy or a cesarean scar pregnancy. Second confusing situation is that the bladder invasion could not be evaluated clearly because a sac location was very close to the bladder. MRI demonstrated that the gestational sac was protruding out of the myometrium at isthmoservical junction at the level of cesarean scar and a sac location was very close to the bladder but it is understood that there was no bladder invasion by monitoring intact bladder wall. In light of these findings, medical therapy was started safely. However, as a result of inadequate medical treatment D & C was performed to the patient.
There is no universal method or consensus for the treatment of cesarean scar pregnancy. The patient’s age, clinical status, pregnancy size, future pregnancy expectations, whether there is a scar rupture, β-hCG levels, technical equipment and the team’s experience are important factors for the choice of treatment. In order to avoid uterine rupture and hemorrhage of the uterus and to preserve fertility and uterus, medical or combined medical and surgical treatment modalities are preferred. Medical treatment is administered systemically or locally. Ultrasound guided injection of methotrexate or potassium chloride to the gestational sac and aspiration of the sac are usually used for local application [11, 12]. Intramuscular methotrexate (1 mg/kg) is applied to patients in systemic medical therapy. Surgical treatment is introduced in cases unresponsive to medical therapy. After medical therapy, follow-up weekly β-hCG should be done until β-hCG level is reached undetectable level and monthly ultrasound follow-up examination should be done until no detection of pregnancy materials. The β-hCG generally reaches its normal levels after 4 - 16 weeks [13]. Gestational sac can be removed by laparotomy or laparoscopy in the surgical treatment. Hysterectomy can be performed in patient with an uncontrolled bleeding and an absence of expected future pregnancies. Another treatment method is uterine artery embolization (UAE). UAE might be a life-saving practice as an alternative to surgical treatment in patients with severe vaginal bleeding and who want to preserve fertility. However, it cannot be performed at all centers because it requires an experienced team and technical equipment [14].
Consequently, the MRI application in cesarean scar pregnancies reveals localization of gestational sac and its relationship with adjacent organs more clearly. Therefore, it provides very important information in determining the treatment plan and guidance the surgeon. As our case report, it is recommended that MRI should be used in addition to TVUS in cesarean scar pregnancy in selected cases that cannot be diagnosed by transvaginal ultrasonography.
| References | ▴Top |
- Ash A, Smith A, Maxwell D. Caesarean scar pregnancy. BJOG. 2007;114(3):253-263.
doi pubmed - Seow KM, Cheng WC, Chuang J, Lee C, Tsai YL, Hwang JL. Methotrexate for cesarean scar pregnancy after in vitro fertilization and embryo transfer. A case report. J Reprod Med. 2000;45(9):754-757.
pubmed - Vial Y, Petignat P, Hohlfeld P. Pregnancy in a cesarean scar. Ultrasound Obstet Gynecol. 2000;16(6):592-593.
doi pubmed - Pang YP, Tan WC, Yong TT, Koh PK, Tan HK, Ho TH. Caesarean section scar pregnancy: a case series at a single tertiary centre. Singapore Med J. 2012;53(10):638-642.
pubmed - Sadeghi H, Rutherford T, Rackow BW, Campbell KH, Duzyj CM, Guess MK, Kodaman PH,
et al . Cesarean scar ectopic pregnancy: case series and review of the literature. Am J Perinatol. 2010;27(2):111-120.
doi pubmed - Seow KM, Huang LW, Lin YH, Lin MY, Tsai YL, Hwang JL. Cesarean scar pregnancy: issues in management. Ultrasound Obstet Gynecol. 2004;23(3):247-253.
doi pubmed - Ofili-Yebovi D, Ben-Nagi J, Sawyer E, Yazbek J, Lee C, Gonzalez J, Jurkovic D. Deficient lower-segment Cesarean section scars: prevalence and risk factors. Ultrasound Obstet Gynecol. 2008;31(1):72-77.
doi pubmed - Armstrong V, Hansen WF, Van Voorhis BJ, Syrop CH. Detection of cesarean scars by transvaginal ultrasound. Obstet Gynecol. 2003;101(1):61-65.
doi - Jurkovic D, Jauniaux E, Kurjak A, Hustin J, Campbell S, Nicolaides KH. Transvaginal color Doppler assessment of the uteroplacental circulation in early pregnancy. Obstet Gynecol. 1991;77(3):365-369.
pubmed - Wu R, Klein MA, Mahboob S, Gupta M, Katz DS. Magnetic resonance imaging as an adjunct to ultrasound in evaluating cesarean scar ectopic pregnancy. J Clin Imaging Sci. 2013;3:16.
doi pubmed - Seow KM, Wang PH, Huang LW, Hwang JL. Transvaginal sono-guided aspiration of gestational sac concurrent with a local methotrexate injection for the treatment of unruptured cesarean scar pregnancy. Arch Gynecol Obstet. 2013;288(2):361-366.
doi pubmed - Ugurlucan FG, Bastu E, Dogan M, Kalelioglu I, Alanya S, Has R. Management of cesarean heterotopic pregnancy with transvaginal ultrasound-guided potassium chloride injection and gestational sac aspiration, and review of the literature. J Minim Invasive Gynecol. 2012;19(5):671-673.
doi pubmed - Jurkovic D, Hillaby K, Woelfer B, Lawrence A, Salim R, Elson CJ. First-trimester diagnosis and management of pregnancies implanted into the lower uterine segment Cesarean section scar. Ultrasound Obstet Gynecol. 2003;21(3):220-227.
doi pubmed - Pyra K, Wozniak S, Szkodziak P, Paszkowski T, Sojk M, Szczerbo-Trojanowska M. [Uterine artery embolisation in massive vaginal bleeding in ectopic pregnancy—case report]. Przegl Lek. 2012;69(7):399-401.
pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


