| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 4, Number 10, October 2013, pages 649-651
Does Bevacizumab Cause Delayed Thromboembolism? Two Cases
Nisar Ahmada, Olugbenga Olowokureb, Sajjad Haidera, Nagla Abdel Karimb, c
aDepartment of Internal Medicine, University of Cincinnati College of Medicine, Cincinnati, OH 45267, USA
bDivision of Hematology and Oncology, University of Cincinnati College of Medicine, Cincinnati, OH 45267, USA
cCorresponding author: Nagla Abdel Karim, 231 Albert Sabin Way, Division of Hematology and Oncology, University of Cincinnati College of Medicine, Cincinnati, OH 45267, USA
Manuscript accepted for publication August 20, 2013
Short title: Bevacizumab Cause Delayed Thromboembolism?
doi: https://doi.org/10.4021/jmc1464w
| Abstract | ▴Top |
Cancer patients are at increased risk of venous thromboembolism (VTE) and this risk may be further increased due to several other factors including chemotherapeutic drugs. Bevacizumab is a vascular endothelial growth factor (VEGF) inhibitor that has been reported to cause arterial and venous thrombosis during the treatment period. We present two cases of venous thrombosis occurring several weeks after stopping bevacizumab, raising the question of a delayed adverse drug effect due to the long half -life of the drug and immune complex formation.
Keywords: Bevacizumab; Thromboembolism; Immune complexes
| Introduction | ▴Top |
Venous thromboembolism (VTE) is responsible for approximately 300,000 deaths annually in US and 20% of these deaths occur among cancer patients [1]. Annual incidence of VTE is estimated to be between 0.5% and 20% depending on the cancer type and background risk [2]. In a population-based case-control study, patients with hematologic malignancies had the highest risk of VTE, followed by patients with lung and gastrointestinal cancers [3]. Cancer patients often have a hypercoagulable state due to the production of substances with procoagulant activity like cancer procoagulant, proinflammatory and proangiogenic cytokines and upregulation of tissue factor expression on tumor cells, endothelial cells, and circulating monocytes [4]. The risk of venous thrombosis in cancer patients is also increased due to various other factors including chemotherapeutic drugs, surgery, hospitalization and placement of central venous catheters [5].
Bevacizumab is a vascular endothelial growth factor (VEGF) inhibitor that has improved survival rates among patients with cancers of lung, colon and glioblastomas. Bevacizumab has been associated with arterial and venous thromboembolism occurring during the treatment period [6, 7]. We describe two cases of venous thrombosis that occurred several weeks after stopping bevacizumab, raising the question of a delayed adverse drug effect.
| Case Report | ▴Top |
Case 1
A 72-year-old female initially presented to an outside hospital with right upper quadrant abdominal pain for 3 weeks. Her past medical history was significant for controlled hypertension and hyperlipidemia. Her family history was significant for heart failure and stroke. She was worked up with a CT scan of the abdomen which showed extensive hepatic metastatic disease, minimal ascites and right mesenteric pericolonic lymphadenopathy. A colonoscopy was performed that showed an ulcerated near-obstructing 6 cm mass in the proximal ascending colon. Biopsies confirmed poorly differentiated adenocarcinoma of the colon. She was referred to our institution for further care. Patient was started on 5-Fluorouracil, oxaliplatin, leucovorin (mFOLFOX6) and bevacizumab. She tolerated chemotherapy very well. However, bevacizumab was discontinued after 2nd cycle of chemotherapy due to wound dehiscence with some drainage at the port site.
Two months later, the patient started having worsening shortness of breath and pleuritic chest pain. She was seen in the clinic. Her Physical examination was unremarkable except for 2 + pedal edema. A CT scan of the chest showed acute pulmonary embolism involving the right upper lobe and posterior segment of the right lower lobe pulmonary arteries. CT scan of the chest did not reveal any pulmonary metastasis and a CT scan of the abdomen later on, revealed stable appearance of thickening of the cecum, hepatic and nodal metastatic disease. Patient was admitted to the hospital for further management with anticoagulation therapy.
Case 2
A 48-year-old female presented to emergency department with chest pain and dyspnea. She had a cardiac workup and was diagnosed with non-ST elevation myocardial infarction (NSTEMI) which was treated medically with heparin drip for 72 hours and aspirin. A CT scan of the chest was also obtained and was negative for pulmonary embolism but showed a 1.6 cm right upper lobe nodule with hilar adenopathy. She underwent bronchoscopy and hilar lymph node biopsy came back positive for adenocarcinoma of the lung. A PET/CT showed uptake in right upper lobe and right hilar and right mediastinal lymphadenopathy. A mediastinoscopy was performed and pathology revealed multiple positive mediastinal lymph nodes. The patient was initially treated with chemo-radiation (cisplatin and etoposide) but progressed. She was then treated with a combination of carboplatin, taxol and bevacizumab for 4 cycles which she tolerated very well and achieved a partial remission. She was then started on maintenance bevacizumab. Bevacizumab was discontinued after 4 months due to intolerance.
Three months later, the patient presented with dyspnea and chest pain and was found to have an acute pulmonary embolism. She was treated with enoxaparin with a questionable compliance. About two months later she also developed neck swelling and CT scan of the neck demonstrated a right internal jugular vein thrombosis. One month later, the patient reported recurrent headaches and MRI of the brain showed thrombosis of the superior sagittal sinus, right sigmoid sinus, right jugular bulb, and upper right internal jugular vein (Fig. 1). MRI of the brain did not show any conclusive evidence of metastasis. CT scan of the chest showed stable appearance of lung mass and lymph nodes and bilateral malignant pleural effusions.
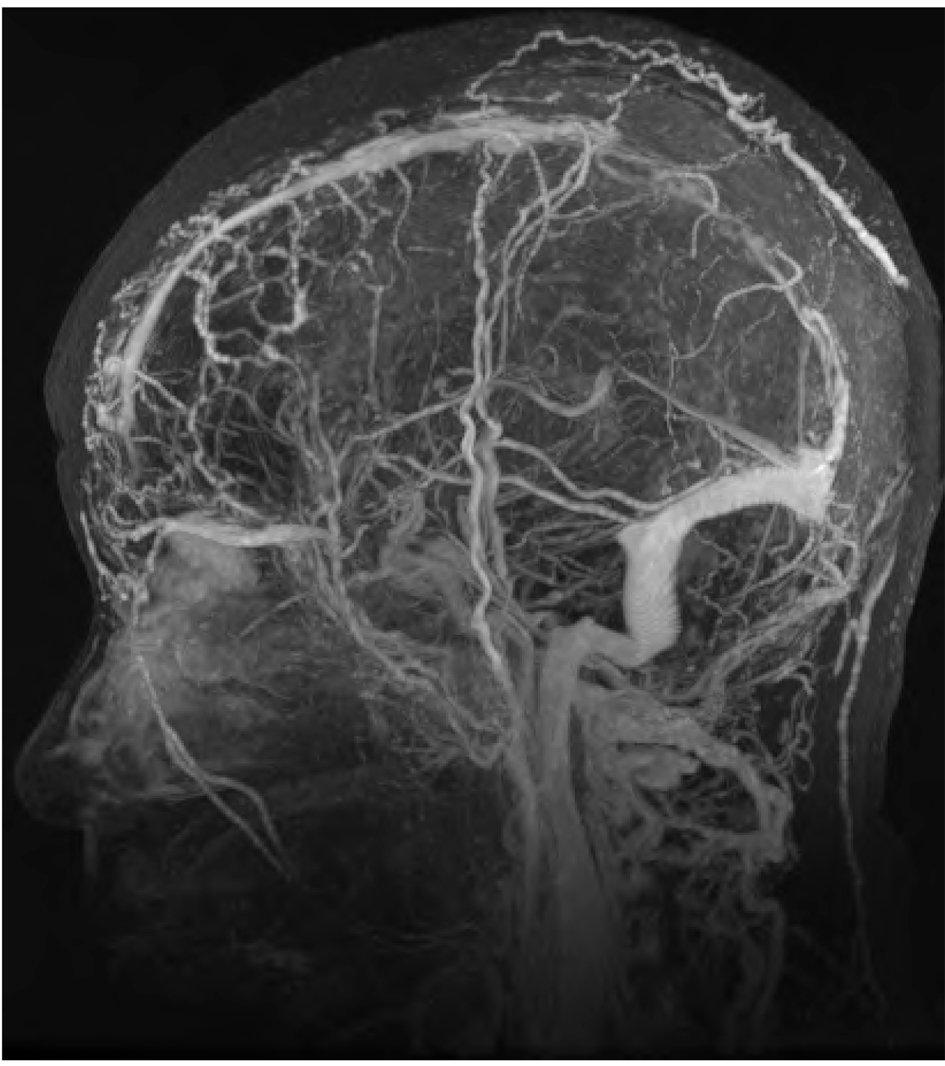 Click for large image | Figure 1. MRI brain showing filling defect within the sagittal sinus on post contrast images. |
| Discussion | ▴Top |
Bevacizumab is a monoclonal antibody that binds to free VEGF and forms immune complexes (IC). Vascular endothelial growth factor is involved in cytoprotection, proliferation of endothelial cells, and increased synthesis of nitric oxide and prostacyclin [8, 9].
Bevacizumab prevents VEGF ligand-receptor binding and down-streams signaling [10]. Anti-VEGF therapy inhibits the development of new tumor vasculature and causes the normalization of surviving tumor vasculature and the regression of existing immature tumor vasculature [11, 12]. Bevacizumab has been reported to cause arterial thrombosis, recurrent PRES (posterior reversible encephalopathy syndrome and venous thromboembolism [7, 13]. Shobha Rani Nalluri et al performed a meta-analysis of 7,956 patients from 15 phase II and II randomized clinical trial (bevacizumab, n = 4,292; control, n = 3,664) and concluded that bevacizumab increased the risk of VTE, both with high and low doses and the risk may vary with tumor type [7].
Bevacizumab related/induced thrombosis may be caused by several mechanisms. First, VEGF inhibition due to bevacizumab results in vascular endothelial cell dysfunction which exposes sub-endothelial procoagulant phospholipids and tissue factor, reduces production of nitric oxide and prostacyclin and increases expression of proinflammatory cytokines [14-16]. All these factors enhance coagulation and thrombosis. In addition, bevacizumab forms immune complexes (IC) with VEGF. These IC can directly induce platelet aggregation and granule release in vitro and cause thrombocytopenia and thrombosis in vivo [14]. While some bevacizumab treated patients may not experience thrombosis during the treatment period, it is possible that cumulative, localized IC assembly and platelet interactions may occur over an extended time period due to the long half-life of the drug (about 20 days; range: 11 - 50 days) [14, 15]. This may explain a delayed effect resulting in thrombosis even after stopping bevacizumab. Finally, VEGF is a physiological regulator of both adult erythropoiesis and synthesis of erythropoietin. Inhibition of VEGF induces normally dormant adult hepatic expression of erythropoietin which may cause thrombosis due to increase in hematocrit and blood viscosity [17].
Our patients presented with thrombotic complications several weeks after stopping bevacizumab. Our patients were at increased risk of thrombosis due to cancer and its progression but bevacizumab may also have contributed to VTE even after discontinuation. We propose that VTE in these patients could have been related to the relatively long half-life of bevacizumab, IC formation and platelet interaction as described above.
Conclusion
Bevacizumab treated cancer patients are at increased risk of VTE and this risk may vary with tumor type. Bevacizumab induced thrombosis usually presents as an acute complication during the treatment period. Here we present two patients experiencing VTE several weeks after stopping the drug. We speculate that VTE in these patients could be a delayed drug adverse effect related to bevacizumab. A long term follow up of bevacizumab treated patients to assess for delayed side effects and analysis of data from various studies and trials involving bevacizumab may be useful to understand the true risk and etiology of such events.
| References | ▴Top |
- Spencer FA, Emery C, Lessard D, Anderson F, Emani S, Aragam J, Becker RC, et al. The Worcester Venous Thromboembolism study: a population-based study of the clinical epidemiology of venous thromboembolism. J Gen Intern Med. 2006;21(7):722-727.
doi pubmed - Horsted F, West J, Grainge MJ. Risk of venous thromboembolism in patients with cancer: a systematic review and meta-analysis. PLoS Med. 2012;9(7):e1001275.
doi pubmed - Khorana AA, Connolly GC. Assessing risk of venous thromboembolism in the patient with cancer. J Clin Oncol. 2009;27(29):4839-4847.
doi pubmed - Prandoni P, Falanga A, Piccioli A. Cancer and venous thromboembolism. Lancet Oncol. 2005;6(6):401-410.
doi - Young A, Chapman O, Connor C, Poole C, Rose P, Kakkar AK. Thrombosis and cancer. Nat Rev Clin Oncol. 2012;9(8):437-449.
doi pubmed - Schutz FA, Je Y, Azzi GR, Nguyen PL, Choueiri TK. Bevacizumab increases the risk of arterial ischemia: a large study in cancer patients with a focus on different subgroup outcomes. Ann Oncol. 2011;22(6):1404-1412.
doi pubmed - Nalluri SR, Chu D, Keresztes R, Zhu X, Wu S. Risk of venous thromboembolism with the angiogenesis inhibitor bevacizumab in cancer patients: a meta-analysis. JAMA. 2008;300(19):2277-2285.
doi pubmed - Senger DR. Vascular endothelial growth factor: much more than an angiogenesis factor. Mol Biol Cell. 2010;21(3):377-379.
doi pubmed - Zachary I. Signaling mechanisms mediating vascular protective actions of vascular endothelial growth factor. Am J Physiol Cell Physiol. 2001;280(6):C1375-1386.
pubmed - Costagliola C, Agnifili L, Arcidiacono B, Duse S, Fasanella V, Mastropasqua R, Verolino M, et al. Systemic thromboembolic adverse events in patients treated with intravitreal anti-VEGF drugs for neovascular age-related macular degeneration. Expert Opin Biol Ther. 2012;12(10):1299-1313.
doi pubmed - Baluk P, Hashizume H, McDonald DM. Cellular abnormalities of blood vessels as targets in cancer. Curr Opin Genet Dev. 2005;15(1):102-111.
doi pubmed - Osusky KL, Hallahan DE, Fu A, Ye F, Shyr Y, Geng L. The receptor tyrosine kinase inhibitor SU11248 impedes endothelial cell migration, tubule formation, and blood vessel formation in vivo, but has little effect on existing tumor vessels. Angiogenesis. 2004;7(3):225-233.
doi pubmed - Lazarus M, Amundson S, Belani R. An Association between Bevacizumab and Recurrent Posterior Reversible Encephalopathy Syndrome in a Patient Presenting with Deep Vein Thrombosis: A Case Report and Review of the Literature. Case Rep Oncol Med. 2012;2012:819546.
- Meyer T, Robles-Carrillo L, Robson T, Langer F, Desai H, Davila M, Amaya M, et al. Bevacizumab immune complexes activate platelets and induce thrombosis in FCGR2A transgenic mice. J Thromb Haemost. 2009;7(1):171-181.
doi pubmed - Verheul HM, Lolkema MP, Qian DZ, Hilkes YH, Liapi E, Akkerman JW, Pili R, et al. Platelets take up the monoclonal antibody bevacizumab. Clin Cancer Res. 2007;13(18 Pt 1):5341-5347.
doi pubmed - Zangari M, Fink LM, Elice F, Zhan F, Adcock DM, Tricot GJ. Thrombotic events in patients with cancer receiving antiangiogenesis agents. J Clin Oncol. 2009;27(29):4865-4873.
doi pubmed - Tam BY, Wei K, Rudge JS, Hoffman J, Holash J, Park SK, Yuan J, et al. VEGF modulates erythropoiesis through regulation of adult hepatic erythropoietin synthesis. Nat Med. 2006;12(7):793-800.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


