| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 4, Number 7, July 2013, pages 447-449
Massive Hemothorax Following Thrombolysis With rtPA in a Stroke Patient With Undiagnosed Rib Fractures
Tomas Belzuneguia, b, c, Carlos Beaumonta, Clint Jean Louisa, Ana Cabodevillaa
aAccident and Emergency Department, Hospital of Navarre, Spain
bDepartment of Health, Public University of Navarre, Spain
cCorresponding author: Tomas Belzunegui, Accident and Emergency Department, Hospital of Navarre, Spain
Manuscript accepted for publication April 24, 2013
Short title: Hemothorax Following Thrombolysis With rtPA
doi: https://doi.org/10.4021/jmc1250w
| Abstract | ▴Top |
The majority of strokes are of ischaemic origin. Thrombolysis has led to a radical change in the outcome of these patients as far as management and prognosis. The main objective is the opening up of the occluded artery and early reperfusion of brain tissue. Only 2% to 4% of patients with acute ischemic stroke receive thrombolytic therapy resulting from the current strict inclusion criteria among other issues. Safety of intravenous and intraarterial thrombolysis in off-label situations is controversially discussed. The most feared complication related to the use of rtPA is intracerebral haemorrhage. Our case is an 84-year-old man who after receiving rtPA in the acute phase of a stroke, presented a massive hemothorax secondary to rib fractures which had not been previously diagnosed at admission. The neurological course was excellent and the hemothorax was resolved after inserting chest drainage. To obtain a meaningful database, a registry for off-label thrombolysis should be created.
Keywords: Thrombolysis; Stroke; Hemothorax; Rib fractures
| Introduction | ▴Top |
Treatment of ischemic stroke dramatically changed after the introduction of thrombolytic therapy in 1995. However, only 2% to 4% of the patients receive intravenous thrombolysis mainly as a result of the short time window and the strict inclusion criteria. As experience with tissue plasminogen activator (tPA) increases, new situations emerge in which the safety of thrombolysis remains unknown. Moreover, the inadvertent or deliberate off-label use of thrombolysis has led to a requestioning of some of the stated exclusion criteria. In these 10 years of experience with tPA, many articles have been published on doubtful situations in which patients were treated.
| Case Report | ▴Top |
An 84-year-old man was brought into the emergency department by the prehospital emergency services after falling down in relation to a sudden onset of weakness in both left extremities. The patient arrived 50 minutes after the onset of symptoms. His vital signs were: blood pressure 158/76 mmHg, heart rate 76 beats per minute (bpm) and capilary glycemia 129. His neurological examination revealed deviation of right labial commissure, dysarthria, hypoesthesia and paresia in the left arm and leg (2/5 in the arm and 3/5 in the leg), with left Babinski and a NIHSS of 12.
The EKG showed sinus rhythm at 80 bpm. A head CT scan ruled out hemorrhage. Doppler studies of the supra-aortic trunk showed normal carotid flow, while transcranial doppler studies revealed an occlusion (TIBI 1 pattern) of the right middle cerebral artery (Fig. 1).
 Click for large image | Figure 1. Systolic spikes of variable velocity and duration. Absent diastolic flow during all cardiac cycles. |
Given the time of evolution and the absence of contraindications, endovenous thrombolysis with rtPA was performed, after which clinical improvement was observed (NIHSS: 5) and a change in doppler pattern with the opening up of the occluded artery (TIBI 4, with systolic velocities of 180 - 200 cm/second) (Fig. 2).
 Click for large image | Figure 2. Systolic velocity: 180 - 200 cm/s. |
Transcranial eco-doppler (GE Vivid 7 Pro, GE Medical Systems, Horten Norway) was performed using a 1.5 - 4 MHz transcranial probe from both temporal bone windows.
After two days the patient referred pain in the right costal margin, with no dyspnea. He recalled falling on the day of admission. A chest radiograph revealed 3 rib fractures (Fig. 3) and a massive hemothorax (Fig. 4) requiring chest drainage (Fig. 5). No complications were observed during his clinical course.
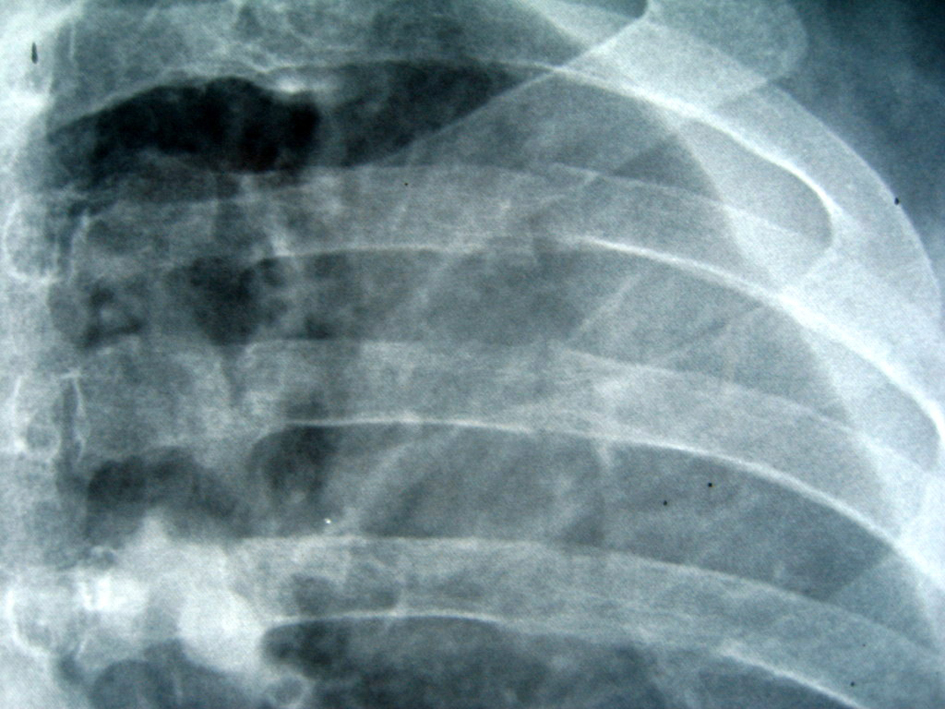 Click for large image | Figure 3. An anteroposterior radiograph shows a radiolucent line without displacement at the posterior portion of three ribs. |
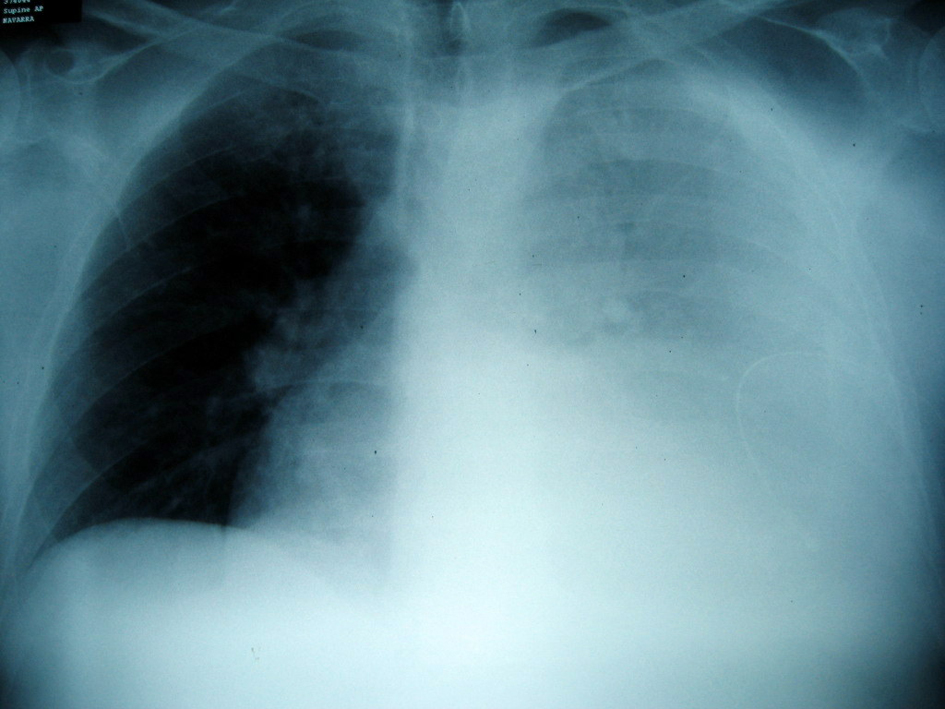 Click for large image | Figure 4. Posteroanterior chest radiograph at presentation with the hemothorax. A large left pleural effusion is prominent. |
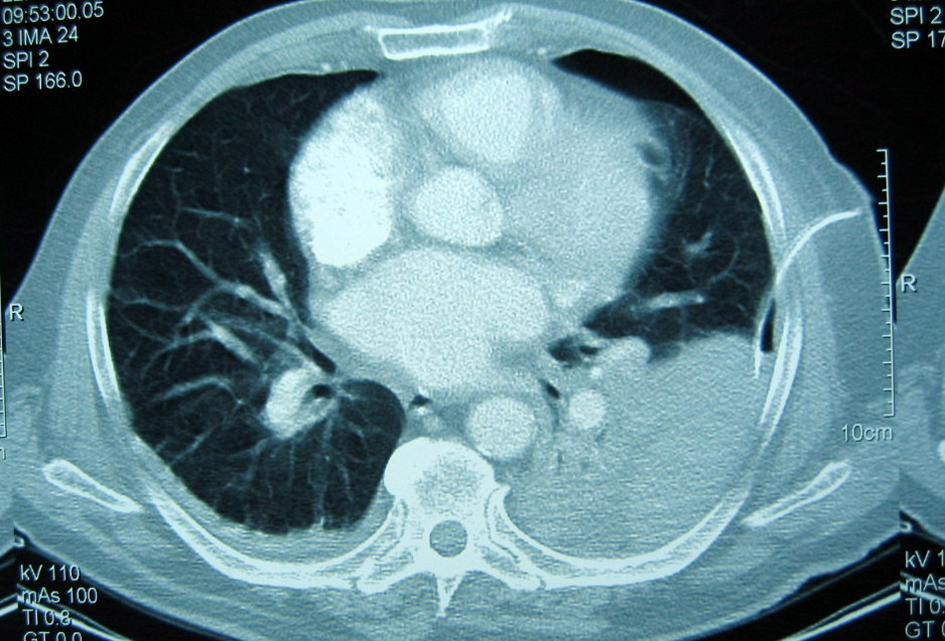 Click for large image | Figure 5. CT scan shows the left hemothorax and the chest drainage. |
On discharge the patient’s NIHSS: 0 points, with only slight limitation in his left hand (Rankin 1 Barthel 85).
| Discussion | ▴Top |
The majority of strokes are of ischaemic origin provoked by a thrombus lodged within an intracraneal artery. Thrombolysis reduces the proportion of dead patients or patients dependent for activities of daily living [1]. Recombinant tissue plasminogen activator (rt-PA), is the only approved drug indicated in the acute phase of stroke [2].
Treatment with rtPA has proved to be effective and secure in the first three hours after the onset of the stroke, with a reduction in mortality and functional dependency [3].
Not many patients benefit from this agent mainly because of the margin time for treatment. Longer door-to-needle time has been shown to be associated with higher rates of intracerebral hemorrhage [4]. Therefore it is important to carefully select candidates for thrombolysis. Exclusion criteria attempt to minimize the risk of bleeding.
Different haemorrhagic related complications have been described in relation to treatment with rtPA [5-10]. Among the requirements for treatment, the patient should show no evidence of active bleeding or acute trauma (fracture) on examination [2]. In our case there was no clinical evidence of trauma (the initial physical examination was normal and the patient did not refer pain or dyspnea). Thus thrombolysis was administered despite the rib fractures which remained undiagnosed initially.
The patient’s neurological evolution was excellent, despite suffering from the hemothorax which was resolved with a chest drain. Should the fracture have been diagnosed beforehand the patient would surely not have received the undeniably beneficial outcome following thrombolysis.
From this we do not want to suggest that the presence of a rib fracture should not be considered as criteria for exclusion from thrombolytic therapy, as it is a potentially severe complication. We simply want to share our experience give the particular outcome of this patient. When therapy with rtPA was initiated for acute stroke, the contraindications were more restrictive and with time and experience there has been an attempt to increase the number of patients who could benefit from it [11]: for instance, the increase in the margin for therapy to more than three hours [12, 13] and its application in patients above 90 years of age [14].
Conclusion
Selected patients might benefit, however, from thrombolysis in situations not currently considered in the inclusion criteria [15].
| References | ▴Top |
- Wardlaw JM, Murray V, Berge E, Del Zoppo GJ. Thrombolysis for acute ischaemic stroke. Cochrane Database Syst Rev. 2009;(4):CD000213.
- Adams HP. Thrombolytics in acute ischaemic stroke: a guide to patient selection and optimum use. BioDrugs. 2000;13(2):115-126.
doi pubmed - Wahlgren N, Ahmed N, Eriksson N, Aichner F, Bluhmki E, Davalos A, Erila T, et al. Multivariable analysis of outcome predictors and adjustment of main outcome results to baseline data profile in randomized controlled trials: Safe Implementation of Thrombolysis in Stroke-MOnitoring STudy (SITS-MOST). Stroke. 2008;39(12):3316-3322.
doi pubmed - Fiorelli M, Alperovitch A, Argentino C, Sacchetti ML, Toni D, Sette G, Cavalletti C, et al. Prediction of long-term outcome in the early hours following acute ischemic stroke. Italian Acute Stroke Study Group. Arch Neurol. 1995;52(3):250-255.
doi pubmed - Hayashi H, Kawamata H, Ichikawa K, Tajima H, Kumazaki T. Rupture of a thoracic aortic aneurysm: a rare adverse reaction following systemic tissue plasminogen activator infusion. Heart Vessels. 2004;19(4):208-211.
doi pubmed - Khawly JA, Ferrone PJ, Holck DE. Choroidal hemorrhage associated with systemic tissue plasminogen activator. Am J Ophthalmol. 1996;121(5):577-578.
pubmed - Kim SH, Choi SH, Song EC, Rha JH, Kim SR, Park HC. Spinal subdural hematoma following tissue plasminogen activator treatment for acute ischemic stroke. J Neurol Sci. 2008;273(1-2):139-141.
doi pubmed - Kremen SA, Wu MN, Ovbiagele B. Hemopericardium following intravenous thrombolysis for acute ischemic stroke. Cerebrovasc Dis. 2005;20(6):478-479.
doi pubmed - Shaps HJ, Snyder GE, Sama AE, Rudolph GS. Airway compromise secondary to lingual hematoma complicating administration of tissue plasminogen activator for acute ischemic stroke. Ann Emerg Med. 2001;38(4):447-449.
doi pubmed - Yip TR, Demaerschalk BM. Forearm compartment syndrome following intravenous thrombolytic therapy for acute ischemic stroke. Neurocrit Care. 2005;2(1):47-48.
doi - De Keyser J. Thrombolytic therapy for acute ischemic stroke. Neurology. 1997;49(3):900-901.
doi pubmed - Del Zoppo GJ, Saver JL, Jauch EC, Adams HP, Jr. Expansion of the time window for treatment of acute ischemic stroke with intravenous tissue plasminogen activator: a science advisory from the American Heart Association/American Stroke Association. Stroke. 2009;40(8):2945-2948.
doi pubmed - Hacke W, Kaste M, Bluhmki E, Brozman M, Davalos A, Guidetti D, Larrue V, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359(13):1317-1329.
doi pubmed - Mateen FJ, Nasser M, Spencer BR, Freeman WD, Shuaib A, Demaerschalk BM, Wijdicks EF. Outcomes of intravenous tissue plasminogen activator for acute ischemic stroke in patients aged 90 years or older. Mayo Clin Proc. 2009;84(4):334-338.
pubmed - Aleu A, Mellado P, Lichy C, Kohrmann M, Schellinger PD. Hemorrhagic complications after off-label thrombolysis for ischemic stroke. Stroke. 2007;38(2):417-422.
doi pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


