| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 13, Number 9, September 2022, pages 438-442
Development of Early Abdominal Compartment Syndrome Leading to Emergent Decompressive Laparotomy in Full-Thickness Burn Injury
Alexandra Nguyena, d, Catherine Trana, Aldin Malkoca, Vivian Davisa, b, Michael M. Neekia, b, c
aDepartment of Surgery, Arrowhead Regional Medical Center, Colton, CA, USA
bCalifornia University of Science and Medicine, Colton, CA, USA
cDepartment of Emergency Medicine, Arrowhead Regional Medical Center, Colton, CA, USA
dCorresponding Author: Alexandra Nguyen, Department of Surgery, Arrowhead Regional Medical Center, Colton, CA 92324, USA
Manuscript submitted June 14, 2022, accepted August 11, 2022, published online September 28, 2022
Short title: Early Abdominal Compartment Syndrome in Burns
doi: https://doi.org/10.14740/jmc3970
| Abstract | ▴Top |
Burn injuries carry an increased risk of intra-abdominal hypertension and are an independent risk factor for abdominal compartment syndrome (ACS). ACS is most commonly due to large volume resuscitation. The added concern of ACS can complicate resuscitative efforts. Early monitoring for ACS (intra-abdominal pressure > 20 mm Hg with associated new-onset organ dysfunction) and performing prudent decompressive laparotomies are important factors to keep in mind when treating large surface area burn patients. This case report describes the hospitalization of a 60-year-old male who presented with 45% full-thickness (FT) total body surface area (TBSA) and inhalation injury. On arrival to the emergency department (ED), he had received a total of 6 L of intravenous lactate Ringers, and vasopressors were initiated due to hypotension. During the tertiary examination it was noted that there was increased difficulty ventilating the patient, and his abdomen was becoming increasingly distended and tense. His intra-abdominal pressure was measured in the ED and found to be elevated at 32 mm Hg. The findings were suggestive of ACS and a decompressive laparotomy was performed in the ED. Upon entering the abdominal cavity, the abdominal contents extruded through the incision and diffuse venous congestion and gastric distention were noted. Items commonly found in operating rooms (Top-Draper® warmer drape, Kerlix rolls, Jackson-Pratt suction drains, and 3M® Ioban sterile antimicrobial incise drape) were utilized to maintain an open abdomen where abdominal contents could easily be observed and to prevent delay in performing a decompressive laparotomy. Here we describe a patient with 45% FT TBSA and inhalation injuries requiring an emergent decompressive laparotomy for ACS after only 6 L of lactate Ringers were administered. This highlights the importance of early monitoring for ACS and the ease of performing a decompressive laparotomy with commonly found items in the ED and operating rooms.
Keywords: Abdominal compartment syndrome; Burn patient; Emergent decompressive laparotomy
| Introduction | ▴Top |
Burn injuries have long been noted to carry an increased risk of intra-abdominal hypertension (IAH) and are identified as an independent risk factor for abdominal compartment syndrome (ACS) [1]. The increased susceptibility to ACS is multifactorial, with the most common etiology being large-volume resuscitation. Furthermore, large volumes of fluid are often necessary and are standard of care in resuscitating patients with severe deep burn injuries. When combined with the systemic inflammatory response, a large infusion of fluid may lead to significant capillary leakage and “third-spacing” of fluid into the abdomen in the form of tissue edema and ascites [2]. In addition, cases of circumferential thoracic and abdominal wall deep burns can result in reduced abdominal wall compliance further propagating the issue [3].
Collectively, these factors create an increase in intra-abdominal pressure (IAP). While baseline IAP can vary per patient, healthy individuals have been found to have an average IAP of 5 - 6 mm Hg [4]. Variations in baseline IAP are influenced by a variety of medical conditions. Obese patients have higher baseline IAP values, some with values even higher than the threshold for IAH of 12 mm Hg [5]. Liver cirrhosis with ascites [6] and pregnancy [7] have inherently elevated IAP. A meta-analysis review by Hershberger et al recommends the need for abdominal pressure monitoring in burn cases with greater than 30% total burn surface area (TBSA) along with early therapeutic maneuvers such as sedation and paralysis, escharotomies, or changes in fluid management may be performed to prevent ACS [8]. Epidemiological studies conducted in the last decades vastly differ in their report of incidence and prevalence. The prevalence of ACS ranged from 0% to 28% [9]. and mortality for ACS can be as high as 60% as evidenced by higher total and renal Sequential Organ Failure Assessment (SOFA) score, lower renal filtration gradient (FG), repetitive use of mechanical ventilation, and longer intensive care unit (ICU) length of stay [10].
Advancements in burn care and technology have improved the survival rate of patients with large full-thickness burns; however, the added concern of ACS further complicates patients’ resuscitative efforts. In severely burned patients described as TBSA > 20%, the prevalence of ACS is estimated to be 4.1-17%, with 65-75% at further risk of developing IAH without end-organ dysfunction [11]. This case report exemplifies the need for vigilant monitoring of ACS and further describes the ease of performing a decompressive laparotomy (DL) with common emergency department and operating room items. Here we describe a patient in the emergency department with 45% TBSA full-thickness burn and inhalation injuries requiring an emergent DL for ACS after 6 L of resuscitative fluids were administered.
| Case Report | ▴Top |
Investigations
A 60-year-old male presented with a 45% full-thickness burns with inhalation injury after being trapped in a structural fire for a prolonged period of time. The patient presented with second-degree burns to the bilateral anterior upper extremities, right upper and lower extremity, scalp, and entirety of the face, with crusted soot and blood noted in the nares. The patient was initially pulseless with the initiation of advanced cardiac life support (ACLS) until obtaining return of spontaneous circulation (ROSC) with systolic blood pressure in the 70s and sinus tachycardia prior to transport to the local community hospital. Per emergency medical services (EMS) the patient had undergone intubation in the prehospital setting due to having a low Glasgow Coma Scale (GCS). After the initial rapid evaluation at the community hospital, the patient was transported to the regional trauma and burn center. He was given a total of 4 L of intravenous (IV) crystalloids (lactate Ringers) and vasopressors (norepinephrine and vasopressin) that were initiated prior to arrival at the regional burn center.
Vitals upon arrival included a blood pressure of 80/56 mm Hg, heart rate of 65 beats per minute, respiratory rate of 14 per minute, pulse oximetry of 71% on 100% oxygen, the core body temperature of 36.1 °C via foley catheter, and GCS 3. An Extended Focused Assessment with Sonography in Trauma (eFAST) exam was noted to have free fluid in Morrison’s pouch and no lung sliding on the left, requiring an emergent placement of a left-sided thoracostomy tube followed by a left subclavian vein central access and right femoral arterial line placement. Fluid resuscitation continued using the modified Parkland formula and the patient was continued on norepinephrine 16 µg/mL and vasopressin 0.03 units/min to improve mean arterial pressure (MAP), along with an IV infusion of supplemental magnesium and potassium for his electrolyte derangement. Approximately 60 mins after arrival at the burn center, during tertiary examination it was noted that there was increased difficulty ventilating the patient with elevated peak airway pressures, arterial blood gas was significant for a pH of 6.83, PaCO2 of 81 mm Hg, PaO2 of 79 mm Hg, bicarbonate of 13 mEq/L, a base deficit of 22 mEq/L, and a lactate 10 mmol/L, and his abdomen was noted to be distended and tense.
Diagnosis
At this time the patient was given an additional 50 mg of vecuronium intravenously and bladder pressure measurements were found to be elevated at 32 mm Hg. Since the findings were suggestive of ACS, the decision was made to perform bedside DL.
Treatment
Upon making the standard surgical incision to enter the abdominal cavity, abdominal contents extruded through the incision and diffuse venous congestion and gastric distention were noted (Fig. 1a). The existing nasogastric tube was slowly advanced and significant tan-colored output was noted. A Top-Draper® warmer drape ORS-100 (O.R. Solutions, Inc., Chantilly, VA) was secured to the fascial edge using non-absorbable sutures in an interrupted fashion (Fig. 1b). Bilaterally Kerlix rolls were placed along the edges of the fascia alongside suction drains. 3M® Ioban sterile antimicrobial incise drape was then applied to the entire abdominal surface, covering the drape, towel, and suction area. Prior to the DL, the patient had received a total of 6 L of crystalloid and two units of packed red blood cells.
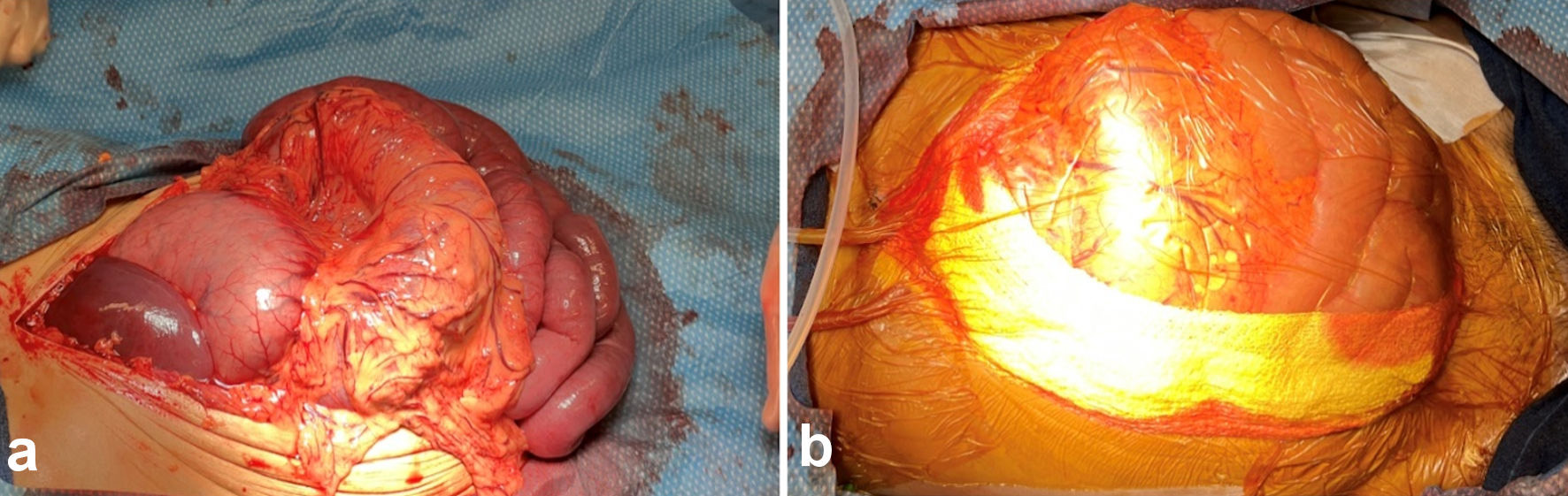 Click for large image | Figure 1. (a) Abdominal contents extruded through midline abdominal incision with venous congestion and gastric distention. (b) Abdominal cavity status post decompressive laparotomy with overlying Top-Draper® warmer drape ORS-100 and Kerlix at bilateral fascial edges with suction drains in place. |
During the stay at the burn unit, the patient’s IAP gradually increased, putting pressure on the covering layer. As a result, the Bogota bag was opened and loosened at the bedside to release the pressure.
Outcomes
No improvement in his neurological exam was noted during the next 48 h and his lactate levels remained elevated accompanied by notable metabolic derangements. During this time, it was noted that the patient had a GCS of 3 with bilateral fixed and dilated pupils and no detectable gag or coughing reflexes. On day 3 of the hospitalization, the patient had an episode of pulseless electrical activity and underwent ACLS protocol resuscitation by the critical care team. ROSC was obtained after one round of resuscitation efforts. Later that day, the patient was determined to have a futile status and was put on the comfort care measures. On his fourth day of admission to the burn unit, the patient succumbed to his injuries.
| Discussion | ▴Top |
ACS is a severe condition caused by increased IAH [12]. We present a 45% full thickness TBSA patient that arrived at the emergency department trauma bay in hypotensive shock and requiring aggressive resuscitation. With minimal fluid resuscitation the patient developed ACS and required a DL in the emergency department highlighting the need for early monitoring and utilization of easy to obtain material to perform a DL to prevent any delay in care.
The consensus and guidelines of IAH and ACS are well defined and summarized by the World Society of the Abdominal Compartment Syndrome (WSACS) [13]. IAP is defined as the pressure within the abdominal cavity with normal IAP ranging from 0 to 5 mm Hg and critically ill patients extending to 7 mm Hg [14]. IAH is defined into four grades categorized by the level of the IAP: grade I is an IAP 12 - 15 mm Hg; grade II is an IAP 16 - 20 mm Hg; grade III is an IAP 21 - 25 mm Hg, and grade IV is an IAP > 25 mm Hg [15]. ACS is usually noted in cases with the IAP greater than 20 mm Hg and is associated with new-onset organ dysfunction [16]. ACS can also be placed into three separated causative categories defined as primary, secondary, and recurrent [16]. Primary ACS occurs as a result of abdominopelvic disease, secondary ACS refers to conditions that do not originate from the abdominopelvic region and recurrent ACS usually reoccurs following previous surgical or medical treatment of primary or secondary ACS [17].
Multiple patient population characteristics, comorbidities, and therapeutic interventions may contribute to the development of IAH and ACS. Hence it is challenging to forecast which patient will eventually develop IAH and ACS. The most common risk factors noted for the development of IAH and/or ACS are large volume crystalloid resuscitation, the respiratory status of the patient, and shock/hypotension [18]. The increase in pressure within the static compartment impairs capillary blood flow, leading to a decrease in arteriole blood flow and decreased venous outflow resulting in cellular hypoxia and anaerobic respiration, and lactic acidosis [16].
Advancements at burn facilities have improved mortality rates in patients with greater than 30% TBSA [19]. Patients with FT burns pose significant challenges including infection, dermal coverage, and management of medical comorbidities. Burn injury disrupts the normal capillary barrier and leads to rapid shifts of intravascular fluid and protein into interstitial spaces. As such, the initial mainstay of treatment in severely burned patients is large volume fluid resuscitation to reduce further cellular injury. While ACS is a rarely reported complication in burn patients the risk of mortality is difficult to determine in the setting of both diseases. Oda et al noted patients requiring 300 mL/kg/24 h were on average 64.5 years of age, had a TBSA of 85%, and the majority of patients had inhalation injury compared to 53.5 years of age, had a TBSA of 37.9%, and no inhalation injury. The modified Baux score takes into consideration TBSA, inhalation injury (inhalation injury is equivalent to an addition of 17 years to a burn patient’s age or 17% to the TBSA), and age to determine the probability of mortality. Modified Baux mortality scores in the group that developed ACS was 95% and the group without ACS was 10%. In these cases, it should be noted that patients with increased modified Baux mortality scores should have close monitoring of peak inspiratory pressure (PIP) and invasive blood pressure (IBP) monitoring to prevent ACS [20].
Fluid resuscitation also may contribute to the development of IAH, a risk that seems especially relevant in patients with capillary leakage, in which fluids accumulate in the bowel wall and mesentery, free peritoneal cavity, retroperitoneum, and abdominal wall [15]. This is especially true for patients requiring high volume resuscitation efforts like in the setting of burn patients. Balancing the need for fluid resuscitation, development of ACS, and treatment of ACS poses extensive difficulties. WSACS management algorithm has five medical treatment arms namely: 1) evacuate intraluminal contents; 2) evacuate intra-abdominal space-occupying lesions; 3) improve abdominal wall compliance; 4) optimize fluid administration; and 5) optimize systemic/regional perfusion [15]. These medical interventions essentially target the three critical contributors to IAH, which are 1) solid-organ and hollow-viscera volume; 2) space-occupying lesions, such as ascites, blood, fluid, or tumors; and 3) conditions that limit the expansion of the abdominal wall [15]. Burn injuries that affect the abdominal and chest wall are thought to affect the expansion of the abdominal wall and due to the pathophysiology of the disease can lead to ascites and third spacing. The unique balance between fluid loss, fluid accumulation, and prevention of ACS is needed to be considered in high-risk burn patients.
In the present study our patient presented in severe shock (systolic blood pressure under 90 mm Hg, base deficit of 22 mEq/L) with severe burn injuries requiring aggressive resuscitation and was on vasopressors. This patient with secondary ACS did not receive resuscitation goals as defined by the modified Parkland formula before the development of ACS. Once surgical decompression was achieved it was noted there was massively and diffusely edematous bowel with present ascites. During the resuscitative efforts, it was noted that the patient continued to exhibit increasingly elevated bladder pressures requiring further abdominal release.
DL has been shown to be effective in decreasing IAP and improving organ function [17]. However, although DL results in a decrease in IAP, IAH has been noted to persist in a considerable number of patients, with mean IAP after DL remaining well above the 12 mm Hg IAH threshold [17]. A study by Balogh et al noted multiple trauma patients undergoing shock resuscitation with increased mortality once patients developed ACS requiring DL [21]. Their findings suggested once patients developed ACS even after DL there was a noted increase in abdominal compartment pressures. This was similarly exhibited by our patient. Therefore, IAP should be closely monitored after DL to prevent recurrent ACS [22].
Learning points
Close monitoring and alternative management of ACS cases need to be part of the regular burn team routine to avoid adverse side effects. Early ACS needs to be considered in patients with severe full-thickness burns and inhalation injury. Additionally, frequent monitoring of bladder pressures is indicated in burn patients during fluid resuscitation to intervene prior to the development of ACS. If needed a DL can be safely performed in an emergency department when needed for ACS.
Acknowledgments
The authors would like to thank and express their gratitude to the Arrowhead Regional Medical Center’s exceptional Emergency Department and burn nursing staff for their expert clinical support.
Financial Disclosure
The research presented in this manuscript had no specific funding from any agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest
None to declare.
Informed Consent
Informed consent was obtained from the patient’s family for publication of this case report and accompanying images, and institutional review board (IRB) approval was granted.
Author Contributions
Alexandra Nguyen, MD wrote the abstract, case, discussion, and added references. Catherine Tran, MD took care of the patient, reviewed, and edited the manuscript. Aldin Malkoc, MD wrote the abstract, patient’s case, discussion, and added figures and references. Vivian Davis, DO, JD took care of the patient, reviewed, and edited the manuscript. Michael M. Neeki, DO, MS took care of the patient, reviewed, and edited the manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
ACS: abdominal compartment syndrome; ACLS: advanced cardiac life support; DL: decompressive laparotomy; eFAST: Extended Focused Assessment with Sonography in Trauma; EMS: Emergency Medical Services; FG: filtration gradient; FT: full-thickness; GCS: Glasgow Coma Scale; IAH: intra-abdominal hypertension; ICU: intensive care unit; IAP: intra-abdominal pressure; IV: intravenous; IBP: invasive blood pressure; MAP: mean arterial pressure; PIP: peak inspiratory pressure; ROSC: return of spontaneous circulation; SOFA: Sequential Organ Failure Assessment; TBSA: total body surface area; WSACS: World Society of the Abdominal Compartment Syndrome
| References | ▴Top |
- Mbiine R, Alenyo R, Kobusingye O, Kuteesa J, Nakanwagi C, Lekuya HM, Kituuka O, et al. Intra-abdominal hypertension in severe burns: prevalence, incidence and mortality in a sub-Saharan African hospital. Int J Burns Trauma. 2017;7(6):80-87.
- Ivy ME, Atweh NA, Palmer J, Possenti PP, Pineau M, D'Aiuto M. Intra-abdominal hypertension and abdominal compartment syndrome in burn patients. J Trauma. 2000;49(3):387-391.
doi pubmed - Blaser AR, Bjorck M, De Keulenaer B, Regli A. Abdominal compliance: A bench-to-bedside review. J Trauma Acute Care Surg. 2015;78(5):1044-1053.
doi pubmed - Frezza EE, Shebani KO, Robertson J, Wachtel MS. Morbid obesity causes chronic increase of intraabdominal pressure. Dig Dis Sci. 2007;52(4):1038-1041.
doi pubmed - De Keulenaer BL, De Waele JJ, Powell B, Malbrain ML. What is normal intra-abdominal pressure and how is it affected by positioning, body mass and positive end-expiratory pressure? Intensive Care Med. 2009;35(6):969-976.
doi pubmed - Umgelter A, Reindl W, Franzen M, Lenhardt C, Huber W, Schmid RM. Renal resistive index and renal function before and after paracentesis in patients with hepatorenal syndrome and tense ascites. Intensive Care Med. 2009;35(1):152-156.
doi pubmed - Lozada MJ, Goyal V, Osmundson SS, Pacheco LD, Malbrain M. It's high time for intra-abdominal hypertension guidelines in pregnancy after more than 100 years of measuring pressures. Acta Obstet Gynecol Scand. 2019;98(11):1486-1488.
doi pubmed - Abdominal compartment syndrome in the severely burned patient. Journal of Burn Care & Research, Oxford Academic. Accessed May 20, 2022. https://academic.oup.com/jbcr/article-abstract/28/5/708/4636840.
doi pubmed - Strang SG, Van Lieshout EM, Van Waes OJ, Verhofstad MH. Prevalence and mortality of abdominal compartment syndrome in severely injured patients: A systematic review. J Trauma Acute Care Surg. 2016;81(3):585-592.
doi pubmed - Abdominal compartment syndrome in the surgical - ProQuest. Accessed May 20, 2022. https://www.proquest.com/docview/212807975?fromopenview=true&pq-origsite=gscholar.
- Henry R, Ghafil C, Golden A, Berry E, Grabo D, Matsushima K, Clark D, et al. A novel abdominal decompression technique to treat compartment syndrome after burn injury. J Surg Res. 2021;260:448-453.
doi pubmed - Talizin TB, Tsuda MS, Tanita MT, Kauss IAM, Festti J, Carrilho C, Grion CMC, et al. Acute kidney injury and intra-abdominal hypertension in burn patients in intensive care. Rev Bras Ter Intensiva. 2018;30(1):15-20.
doi pubmed - Papavramidis TS, Marinis AD, Pliakos I, Kesisoglou I, Papavramidou N. Abdominal compartment syndrome - Intra-abdominal hypertension: Defining, diagnosing, and managing. J Emerg Trauma Shock. 2011;4(2):279-291.
doi pubmed - Malbrain ML, Cheatham ML, Kirkpatrick A, Sugrue M, Parr M, De Waele J, Balogh Z, et al. Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. I. Definitions. Intensive Care Med. 2006;32(11):1722-1732.
doi pubmed - Newman RK, Dayal N, Dominique E. Abdominal Compartment Syndrome. In: StatPearls. Treasure Island (FL), 2022.
- Cheatham ML. Abdominal compartment syndrome: pathophysiology and definitions. Scand J Trauma Resusc Emerg Med. 2009;17:10.
doi pubmed - De Waele JJ, Hoste EA, Malbrain ML. Decompressive laparotomy for abdominal compartment syndrome—a critical analysis. Crit Care. 2006;10(2):R51.
doi pubmed - Holodinsky JK, Roberts DJ, Ball CG, Blaser AR, Starkopf J, Zygun DA, Stelfox HT, et al. Risk factors for intra-abdominal hypertension and abdominal compartment syndrome among adult intensive care unit patients: a systematic review and meta-analysis. Crit Care. 2013;17(5):R249.
doi pubmed - Malkoc A, Wong DT. Lessons learned from two survivors of greater than 90% TBSA full-thickness burn injuries using novosorb biodegradable temporizing matrix and autologous skin cell suspension, RECELL: a case Series. J Burn Care Res. 2021;42(3):577-585.
doi pubmed - Oda J, Yamashita K, Inoue T, Harunari N, Ode Y, Mega K, Aoki Y, et al. Resuscitation fluid volume and abdominal compartment syndrome in patients with major burns. Burns. 2006;32(2):151-154.
doi pubmed - Balogh Z, McKinley BA, Cocanour CS, Kozar RA, Holcomb JB, Ware DN, Moore FA. Secondary abdominal compartment syndrome is an elusive early complication of traumatic shock resuscitation. Am J Surg. 2002;184(6):538-543; discussion 543-534.
doi - Duchesne JC, Baucom CC, Rennie KV, Simmons J, McSwain NE, Jr. Recurrent abdominal compartment syndrome: an inciting factor of the second hit phenomenon. Am Surg. 2009;75(12):1193-1198.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


